Japan is one of the few countries in the world where stem cell therapy is both legal and regulated by the national government. Under the Act on the Safety of Regenerative Medicine (ASRM), clinics must submit detailed treatment plans to the Ministry of Health, Labour and Welfare (MHLW) and operate under strict oversight — a level of transparency that most other countries simply do not offer.
But regulation alone does not answer the questions that matter most to patients: Does it actually work? Is it safe? How much does it cost? And how do I know which clinic to trust?
This guide was written to answer those questions — not with marketing claims, but with published clinical evidence. Below, you will find data from randomized controlled trials and meta-analyses, an honest breakdown of costs, and a step-by-step explanation of what the treatment process looks like in Japan.

- 1. What Is Stem Cell Therapy?
- 2. Why Japan? The Regulatory Framework
- 3. What Conditions Can Be Treated?
- 4. The Treatment Process — From Consultation to Recovery
- 5. How Much Does It Cost?
- 6. Is It Safe? Risks and Side Effects
- 7. Japan vs. Other Countries
- 8. How to Choose a Clinic in Japan
- 9. Frequently Asked Questions
- References
1. What Is Stem Cell Therapy?
Stem cell therapy is a form of regenerative medicine that uses the body’s own repair cells to reduce inflammation, promote tissue healing, and restore function in damaged organs. Rather than masking symptoms with medication, it aims to address the underlying biological cause of disease.
How Mesenchymal Stem Cells (MSCs) Work
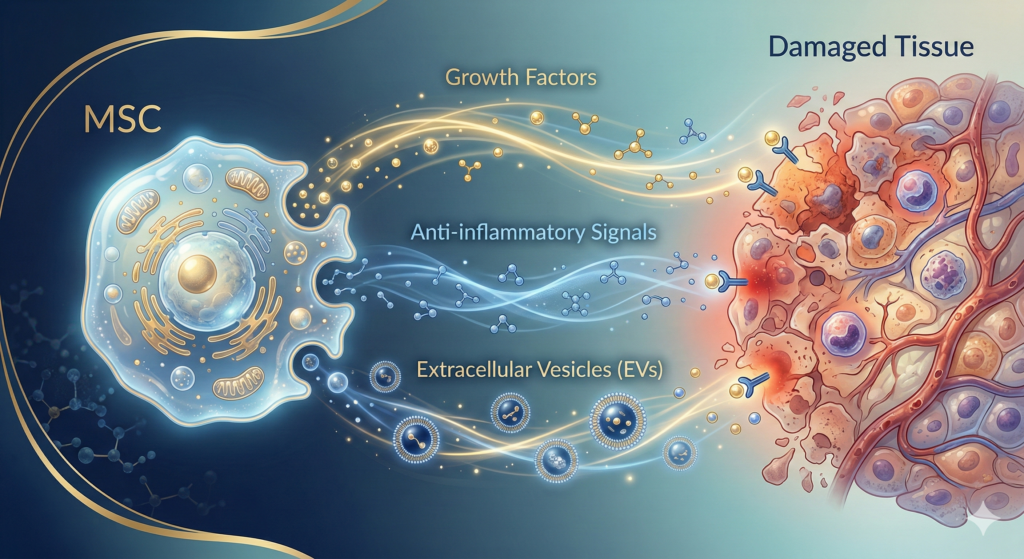
The most widely used cell type in clinical practice is the mesenchymal stem cell, or MSC. These cells are found naturally in fat tissue, bone marrow, and other sources throughout the body. When harvested, multiplied in a laboratory, and reintroduced into the patient, MSCs exert their effects primarily through a mechanism called the paracrine effect — they release hundreds of bioactive molecules (growth factors, cytokines, and extracellular vesicles) that:
- Suppress excessive inflammation
- Stimulate the growth of new blood vessels (angiogenesis)
- Recruit the body’s own repair cells to the site of damage
- Modulate the immune system to prevent it from attacking healthy tissue
It is important to understand that MSCs do not simply “turn into” new cartilage, new neurons, or new heart muscle. Their primary role is to create a healing environment that allows the body’s own tissues to recover. This distinction matters — and it is backed by decades of research.
Autologous vs. Allogeneic — Why the Source of Cells Matters
Autologous stem cells are harvested from the patient’s own body — typically from a small sample of abdominal fat tissue. Because these cells carry your own DNA, there is essentially zero risk of immune rejection. This is the standard approach in Japan’s MHLW-regulated clinics.
Allogeneic stem cells come from a donor — often umbilical cord tissue. While these cells are available “off the shelf” and do not require a harvesting procedure, they carry a theoretical risk of immune response. In Japan, allogeneic therapies face stricter regulatory requirements and are generally limited to clinical trials.
Adipose (fat) tissue is the preferred source for autologous MSCs in Japan because it yields a significantly higher concentration of stem cells compared to bone marrow — and the harvesting procedure is far less invasive (a small sample from the abdomen under local anesthesia, rather than a bone marrow aspiration).
2. Why Japan? The Regulatory Framework
Stem cell therapy exists in a legal grey area in most countries. In the United States, the FDA restricts cultured (expanded) stem cell therapies to clinical trials. In Mexico, Thailand, and parts of Central America, treatments are widely available but often operate without government oversight.
Japan occupies a unique position: it is one of the only countries where cultured autologous stem cell therapy is both legal and strictly regulated under national law.
The Act on the Safety of Regenerative Medicine (ASRM)
Enacted in 2014, the ASRM requires every clinic that provides regenerative medicine to submit a detailed treatment plan — called a Regenerative Medicine Provision Plan — to the MHLW. This plan must be reviewed and approved by an independent, government-certified Special Committee before any patient can be treated.
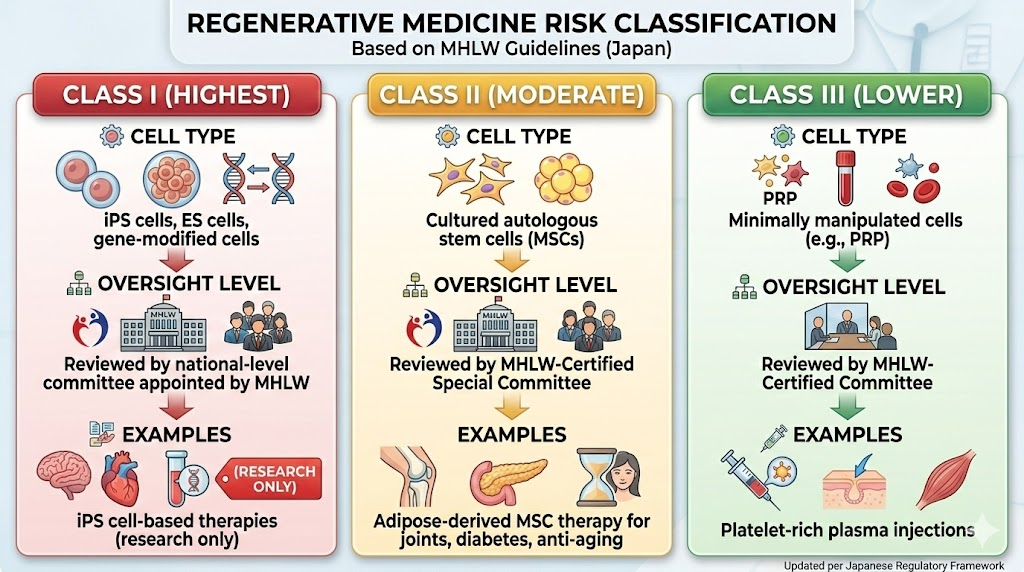
The law classifies regenerative medicine into three risk tiers:
| Risk Class | Cell Type | Oversight Level | Examples |
| Class I (Highest) | iPS cells, ES cells, gene-modified cells | Reviewed by national-level committee appointed by MHLW | iPS cell-based therapies (research only) |
| Class II (Moderate) | Cultured autologous stem cells (MSCs) | Reviewed by MHLW-Certified Special Committee | Adipose-derived MSC therapy for joints, diabetes, anti-aging |
| Class III (Lower) | Minimally manipulated cells (e.g., PRP) | Reviewed by MHLW-Certified Committee | Platelet-rich plasma injections |
Most stem cell treatments offered at private clinics in Japan — including adipose-derived MSC therapy — fall under Class II. This means they have been reviewed by an independent committee, the treatment protocol has been filed with the government, and the clinic is subject to ongoing monitoring and inspection.
Cell Processing Centers (CPCs)
Under Japanese law, stem cells cannot simply be processed in a back room. They must be cultured in a government-licensed Cell Processing Center (CPC) that meets strict standards for sterility, contamination control, and quality assurance. In addition, in-hospital CPCs generally operate under a notification system (filing only), whereas outsourced, external CPCs must obtain formal accreditation as well—meaning they are run under even more stringent operational standards and oversight. Each batch of cells undergoes testing for bacteria, mycoplasma, endotoxins, and viability before it is cleared for administration.
This infrastructure is one of the key differences between Japan and countries where stem cell therapy operates outside a formal regulatory framework.
3. What Conditions Can Be Treated?
Patients often ask: “How can one treatment possibly work for so many different conditions?” It is a fair question — and the answer is important.
Stem cell therapy is not a drug designed to target a single disease. It is a biological mechanism. MSCs reduce inflammation, modulate the immune system, and promote tissue repair — processes that are relevant to virtually every chronic disease in the body. Arthritis, diabetes, heart failure, and neurodegenerative diseases all share a common thread: chronic inflammation and progressive tissue damage.
Think of it this way: a fire extinguisher does not “treat” kitchen fires, electrical fires, and forest fires as three separate diseases. It addresses the underlying problem — fire. Similarly, MSCs address the underlying biology — inflammation and cellular damage — which is why the same cell type can show benefit across seemingly unrelated conditions.
This is not a marketing claim. It is a reflection of the fact that MSCs work through systemic biological mechanisms, not disease-specific drug targets. Below is a summary of the conditions most commonly treated in Japan, along with the level of clinical evidence available for each.
Orthopedic — Knee Osteoarthritis & Chronic Pain
Knee osteoarthritis is the most widely studied application of MSC therapy. A 2025 meta-analysis published in Stem Cell Research & Therapy analyzed 8 randomized controlled trials involving 502 patients and found that MSC injection into the knee joint produced significant improvements in pain and function (WOMAC score) at both 6 and 12 months. Higher cell doses and adipose-derived cells showed the strongest results, with no increase in adverse events compared to controls.
For patients who have tried hyaluronic acid injections, physical therapy, and oral medications without lasting relief — and who want to avoid or delay knee replacement surgery — stem cell therapy offers a non-surgical alternative supported by Level I evidence.
→ Read more: Stem Cell Therapy for Knee Osteoarthritis
Metabolic — Diabetes (Type 1 & Type 2)
A 2025 meta-analysis in Stem Cell Research & Therapy, covering 34 randomized controlled trials, reported that MSC therapy was significantly more effective than placebo for diabetes management (odds ratio 2.79). Autologous MSCs outperformed allogeneic sources. In Type 2 diabetes patients, MSC therapy has been shown to improve HbA1c levels, reduce insulin requirements, and improve beta-cell function.
A separate review in the Journal of Clinical Endocrinology and Metabolism summarized the molecular mechanisms: adipose-derived MSCs reduce pancreatic inflammation, promote beta-cell regeneration, and improve insulin sensitivity through paracrine signaling.
→ Read more: Stem Cell Therapy for Diabetes
Neurological — Stroke Recovery
A Japanese clinical study published in the International Journal of Stem Cell Research & Therapy followed 21 chronic-phase stroke patients who received intravenous autologous adipose-derived stem cells. Early improvements in motor, sensory, and cognitive function were observed, with patients treated within 6 months of onset showing the most pronounced recovery.
A broader meta-analysis of stem cell therapies for ischemic stroke, published in Neurological Sciences (2024), confirmed an overall favorable safety profile, though it noted significant variability between trials — underscoring the importance of standardized protocols and adequate cell doses.
→ Read more: Stem Cell Therapy for Stroke & Cardiovascular Disease
Neurological — Alzheimer’s Disease
In one of the most significant developments in recent years, a 2025 randomized controlled trial published in Nature Medicine tested allogeneic MSC therapy (laromestrocel) in 49 patients with mild Alzheimer’s disease across 10 U.S. centers. The results showed a 48.4% reduction in whole-brain volume loss (P=0.005) and a 61.9% improvement in left hippocampal volume compared to placebo over 39 weeks. While this is still Phase 2a, it represents the first RCT evidence that MSC therapy may slow structural brain degeneration in Alzheimer’s.
Cardiovascular — Heart Failure
A 2024 meta-analysis in the Journal of Translational Medicine, analyzing 17 randomized controlled trials with 1,684 patients, found that MSC transplantation reduced mortality risk by 22% (relative risk 0.78) and significantly improved left ventricular ejection fraction (LVEF +3.38%). Autologous MSCs delivered via coronary artery injection showed the strongest results.
Anti-Aging & Longevity
Age-related frailty is driven by the progressive decline of the body’s regenerative capacity — including reduced stem cell populations, increased systemic inflammation (“inflammaging”), and accumulated cellular damage. MSC therapy aims to partially reverse this process by replenishing anti-inflammatory and pro-regenerative signaling.
A 2021 review in Theranostics outlined the mechanisms by which MSCs address frailty: reducing chronic inflammation, improving immune function, promoting vascular repair, and enhancing tissue regeneration. Clinical trials on aging frailty have shown improvements in physical performance, inflammatory markers, and quality of life.
→ Read more: Back-Aging — Longevity & Anti-Aging
Erectile Dysfunction (ED)
For men whose ED does not respond adequately to PDE5 inhibitors (such as Viagra or Cialis) — particularly those with diabetes-related vascular damage — stem cell therapy targets the root cause: damaged penile blood vessels and smooth muscle tissue. A 2025 review in Frontiers in Medicine summarized preclinical and emerging clinical evidence showing that MSCs promote angiogenesis, nerve regeneration, and smooth muscle repair in the corpus cavernosum.
→ Read more: Stem Cell Therapy for ED
Other Conditions Under Investigation
MSC therapy is also being studied for multiple sclerosis, liver cirrhosis, chronic kidney disease, intervertebral disc degeneration, and autoimmune conditions such as rheumatoid arthritis. Evidence levels vary — some conditions have multiple RCTs, while others remain at the pilot-study stage. Patients should ask their physician about the specific evidence available for their condition.
→ Read more: Stem Cell Therapy for Chronic Pain
Reach us directly — WhatsApp and email inquiries are free of charge.
4. The Treatment Process — From Consultation to Recovery
For international patients, stem cell therapy in Japan typically requires two visits, separated by a cultivation period of several weeks. Here is what to expect at each stage.
Step 1 — Medical Consultation & Screening
Before any treatment begins, a physician conducts a thorough evaluation including medical history review, blood tests, and imaging as needed. This step determines whether stem cell therapy is appropriate for your condition — and it is also where a responsible clinic may advise against treatment if the evidence does not support it for your specific case.
Many clinics offer an initial remote consultation via video call, allowing international patients to discuss their condition before traveling.
Step 2 — Fat Tissue Harvesting (First Visit: 1–2 Days)
A small sample of fat tissue (approximately 1–3 grams) is collected from the abdomen under local anesthesia. The procedure takes about 20–30 minutes and is performed on an outpatient basis — you can walk out of the clinic afterward. Blood samples are also drawn for infection screening (HIV, hepatitis B/C, syphilis, HTLV-1).
Step 3 — Cell Cultivation in a Certified CPC (5-7 Weeks)
This is the stage that separates Japan’s approach from “same-day” procedures offered in some countries. Your stem cells are transported to a government-licensed Cell Processing Center, where they are cultured for approximately 5-7 weeks. During this time:
- The cells are expanded from a small number to 100–200 million or more
- Quality testing is performed at multiple checkpoints (sterility, viability, mycoplasma, endotoxins)
- Only cells that meet strict release criteria are cleared for administration
The 5-7 week cultivation period is not arbitrary — it allows time for the cells to be carefully selected, multiplied, and rigorously tested. “Same-day” procedures, by contrast, typically deliver far fewer cells (often under 10 million) with minimal quality testing.
During this period, you return home and await notification that your cells are ready.
Step 4 — Administration (Second Visit: 1–2 Days)
Depending on the condition being treated, cells are delivered by intravenous (IV) infusion for systemic conditions (diabetes, anti-aging, stroke, heart failure) or by local injection for targeted conditions (knee, joints, ED). The procedure itself is typically completed within 1–2 hours on an outpatient basis.
Step 5 — Follow-Up
Most clinics schedule follow-up assessments at 1, 3, and 6 months post-treatment. Results are not immediate — the biological repair process builds gradually, with most patients noticing meaningful changes between 1 and 6 months after treatment.
Note: Procedures may differ depending on the clinic. Please confirm the details with your attending clinic.

5. How Much Does It Cost?
Why Prices Vary — The Wagyu Analogy
Pricing for stem cell therapy is a bit like ordering steak: you can get a basic cut at a chain restaurant, or you can get A5 Wagyu from Kobe. Both are “steak,” but the quality, sourcing, preparation, and experience are vastly different.
In stem cell therapy, the factors that drive cost include:
- Cell count: A treatment delivering 50 million cells is not comparable to one delivering 200 million cells. Higher counts require longer cultivation and more laboratory resources.
- Culture duration: “Same-day” procedures (no culture) are cheaper but deliver far fewer cells. A 7-week culture with multiple quality checkpoints costs more — but produces a fundamentally different product.
- Quality testing: Sterility testing, viability assays, mycoplasma screening, and endotoxin testing add cost but are essential for safety.
- Regulatory compliance: Operating under MHLW regulation — with CPC certification, committee review, and government reporting — carries overhead that unregulated clinics do not have.
- Physician expertise: Board-certified regenerative medicine specialists with research backgrounds command different fees than general practitioners.
General Price Ranges in Japan
The following are approximate ranges for stem cell therapy in Japan. Actual costs vary by clinic, cell count, condition, and treatment protocol.
| Treatment Type | Approximate Range (USD) | Notes |
| Localized (single joint) | $15,000 – $25,000 | Knee, shoulder, or hip injection |
| Systemic IV (single session) | $20,000 – $40,000 | Diabetes, anti-aging, neurological conditions |
| Comprehensive package | $25,000 – $50,000+ | Multi-session protocols for complex conditions |
| Exosome / supernatant therapy | $1,000 – $10,000 | Cell-free alternative; lower cost, shorter process |
These prices reflect Japan’s premium regulatory environment. They include physician consultation, cell harvesting, CPC cultivation, quality testing, and administration. International patients should also factor in travel, accommodation, and any translation services.
What’s Included (and What Isn’t)
When comparing prices between clinics — or between countries — always ask what is included in the quoted price. Some clinics bundle everything (consultation, harvesting, culture, administration, follow-up) into a single package, while others charge separately for each component. Key questions to ask:
- Does the price include cell cultivation and quality testing?
- How many cells will be administered?
- Are follow-up consultations included?
- Is there an additional fee for the harvesting procedure?
- What happens if the cells fail quality testing — is there a re-culture at no extra charge?
6. Is It Safe? Risks and Side Effects
Safety is the most important consideration for any medical treatment — and it is especially important for therapies that involve living cells.
What Meta-Analyses Say About Safety
A large-scale meta-analysis covering 15 years of MSC clinical trials found no significant increase in serious adverse events compared to control groups. The most commonly reported side effects were mild and transient: low-grade fever, minor pain at the injection or harvesting site, and temporary swelling.
A 2024 systematic review of extracellular vesicle (EV) therapy — a related approach using cell-derived particles rather than whole cells — analyzed 21 clinical reports and found a serious adverse event rate of just 0.7%, with no significant difference in safety between autologous and allogeneic sources.
Common Side Effects vs. Serious Adverse Events
| Category | Examples | Frequency |
| Common (mild) | Low-grade fever, soreness at harvesting site, mild swelling | 10–20% of patients; resolves within days |
| Uncommon | Headache, fatigue, temporary joint stiffness | < 5% of patients |
| Serious | Infection, allergic reaction, blood clot | < 1% in regulated settings; extremely rare with autologous cells |
How Japan’s Regulation Minimizes Risk
The MHLW regulatory framework directly addresses the major safety risks:
- Contamination risk → Mandatory CPC certification with sterility testing at multiple checkpoints
- Unqualified providers → Treatment plans must be reviewed by an independent certified committee
- Lack of follow-up → Clinics must report outcomes and adverse events to the government
- Unknown cell quality → Viability, potency, and contamination testing are required before release
This does not make stem cell therapy risk-free. No medical treatment is. But Japan’s regulatory structure provides a level of safety assurance that is not available in most countries where stem cell therapy is offered.
7. Japan vs. Other Countries
Patients considering stem cell therapy often compare options across multiple countries. The table below summarizes key differences in regulatory environment, cell processing, and typical costs.
| Factor | Japan | South Korea | Mexico | Thailand | United States |
| Government regulation | MHLW / ASRM (specific law) | MFDS (specific law) | COFEPRIS (limited oversight) | Thai FDA (evolving) | FDA (cultured cells restricted to trials) |
| Cultured cells legal? | Yes (Class II) | Yes (approved products) | Grey area | Grey area | No (clinical trials only) |
| Cell source | Autologous (adipose/BM) | Auto + Allo | Varies widely | Varies widely | Research only |
| CPC required? | Yes (government-certified) | Yes (KGMP) | Not required | Varies | FDA cGMP (research) |
| Typical cost range | $15K – $50K | $12K – $30K | $5K – $20K | $8K – $25K | $20K+ (trials only) |
| Cell count (typical) | 100M – 200M+ | 50M – 200M | Varies; often < 50M | Varies | Varies by trial |
| Quality testing mandatory? | Yes | Yes | No | Varies | Yes (trial protocol) |
The key takeaway: Japan and South Korea are the only two countries with dedicated laws specifically regulating regenerative medicine at private clinics. In most other countries, stem cell therapy either operates in a regulatory grey area or is limited to clinical trials.
Lower cost does not necessarily mean lower quality — but it often means less regulatory oversight, fewer quality checkpoints, and less transparency about what you are actually receiving.
8. How to Choose a Clinic in Japan
Not all stem cell clinics in Japan are equal. While MHLW regulation provides a baseline, the quality of treatment varies based on physician expertise, cell processing standards, and transparency. Here are five questions every patient should ask:
1. “What is your MHLW Regenerative Medicine Provision Plan number?”
Every legally operating clinic must have a registered plan number for each specific treatment they offer. If a clinic cannot provide this, it may not be operating within the regulatory framework. You can verify plan numbers through the MHLW database.
2. “How many cells will you administer, and how long is the cultivation period?”
Cell count matters. Research consistently shows that higher doses are associated with better outcomes. Ask whether the clinic delivers 50 million, 100 million, or 200+ million cells — and whether the culture period is measured in days or weeks. A 7-week culture with quality checkpoints is very different from a same-day procedure.
3. “Is your Cell Processing Center (CPC) externally certified?”
The CPC is where your cells spend weeks being multiplied and tested. Ask whether it is an in-house facility or an independent, certified facility — and what quality testing is performed before your cells are released for treatment.
4. “What are the physician’s qualifications and research background?”
Regenerative medicine is a specialized field. Ask whether the treating physician holds board certification in regenerative medicine (such as from the American Board of Regenerative Medicine or the Japanese Society for Regenerative Medicine etc), has published research in peer-reviewed journals, or has training in regenerative medicine field.
5. “Can you show me the published evidence for this treatment?”
A responsible clinic should be able to point you to specific meta-analyses, RCTs, or clinical studies that support the treatment they are recommending — not just patient testimonials or before-and-after photos.

9. Frequently Asked Questions
Is stem cell therapy legal in Japan?
Yes. Stem cell therapy is legal and regulated under the Act on the Safety of Regenerative Medicine (ASRM). Clinics must submit treatment plans to the Ministry of Health, Labour and Welfare (MHLW) and operate under government-certified committee oversight.
How many cells are typically used in a treatment?
Cell counts vary by clinic and condition. In Japan, cultured adipose-derived MSC therapies typically deliver between 100 million and 200+ million cells per session. Same-day, uncultured procedures in other countries may deliver fewer than 10 million — a fundamentally different therapeutic dose.
How long do I need to stay in Japan?
Most protocols require two visits. The first visit (1–2 days) is for consultation and fat tissue harvesting. After approximately 7 weeks of cell cultivation, you return for the second visit (1–2 days) for cell administration. Total time in Japan is typically 3–5 days spread across two trips.
Is stem cell therapy covered by insurance?
In most cases, no. Stem cell therapy in Japan is classified as elective regenerative medicine and is not covered by standard health insurance — either Japanese national insurance or international insurance plans. Clinics provide transparent pricing for self-pay patients.
How soon can I expect results?
Results are not immediate. The biological repair process is gradual. Most patients begin to notice changes between 1 and 3 months after treatment, with peak effects typically observed between 3 and 6 months. Some conditions, such as joint osteoarthritis, may show earlier improvement.
What is the difference between cultured and same-day stem cells?
Same-day (uncultured) procedures process your cells immediately, delivering a relatively small number of cells within hours. Cultured procedures grow your cells over several weeks in a certified laboratory, producing 10–20 times more cells with quality verification at each stage. Japan’s regulatory framework is specifically designed around cultured cell therapies.
Can I store my cells for future use?
Some clinics offer cell banking — freezing a portion of your cultured cells for potential future treatments. Since stem cells decline in number and vitality with age, banking cells at a younger age can preserve their quality for later use.
What should I look for in a clinic’s credentials?
Look for: (1) MHLW Regenerative Medicine Provision Plan registration numbers, (2) Cell Processing Center, (3) physician board certification in regenerative medicine, (4) experience in regenarative medicine, and (5) the ability to cite specific clinical evidence for the treatment
Reach us directly — WhatsApp and email inquiries are free of charge.
References
1. Cao M, et al. Efficacy and safety of mesenchymal stem cells in knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Stem Cell Res Ther. 2025;16(1):122. https://doi.org/10.1186/s13287-025-04252-2
2. Li Y, et al. A meta-analysis on application and prospect of cell therapy in the treatment of diabetes mellitus. Stem Cell Res Ther. 2025;16(1):249. https://doi.org/10.1186/s13287-025-04377-4
3. Mikłosz A, Chabowski A. Adipose-derived Mesenchymal Stem Cells Therapy as a new Treatment Option for Diabetes Mellitus. J Clin Endocrinol Metab. 2023;108(8):1889-1897. https://doi.org/10.1210/clinem/dgad142
4. Kavousi SA, et al. Efficacy of mesenchymal stem cell transplantation on major adverse cardiovascular events and cardiac function indices in patients with chronic heart failure: a meta-analysis of randomized controlled trials. J Transl Med. 2024;22:786. https://doi.org/10.1186/s12967-024-05352-y
5. Rash BG, et al. Allogeneic mesenchymal stem cell therapy with laromestrocel in mild Alzheimer’s disease: a randomized controlled phase 2a trial. Nat Med. 2025;31(4):1257-1266. https://doi.org/10.1038/s41591-025-03559-0
6. Ichihashi Y, et al. Therapeutic Effect of Intravenously Administered Autologous Adipose-Derived Stem Cells on Chronic Stage Stroke Patients. Int J Stem Cell Res Ther. 2020;7:070. https://doi.org/10.23937/2469-570X/1410070
7. Hovhannisyan A, et al. A review and meta-analysis of stem cell therapies in stroke patients: effectiveness and safety evaluation. Neurol Sci. 2024;45:65-74. https://doi.org/10.1007/s10072-023-07032-z
8. Zhu Y, et al. Application of mesenchymal stem cell therapy for aging frailty: from mechanisms to therapeutics. Theranostics. 2021;11(12):5675-5685. https://doi.org/10.7150/thno.46436
9. Fu X, et al. Advances in stem cell therapy for erectile dysfunction: preclinical evidence and emerging therapeutic approaches. Front Med. 2025;12:1519095. https://doi.org/10.3389/fmed.2025.1519095
10. Van Delen S, et al. A systematic review and meta-analysis of clinical trials assessing safety and efficacy of human extracellular vesicle-based therapy. J Extracell Vesicles. 2024;13:e12458. https://doi.org/10.1002/jev2.12458
11. Wang Y, Yi H, Song Y. The safety of MSC therapy over the past 15 years: a meta-analysis. Stem Cell Res Ther. 2021;12(1):545. https://doi.org/10.1186/s13287-021-02609-x
12. Lu W, et al. Efficacy and safety of mesenchymal stem cell therapy in liver cirrhosis: a systematic review and meta-analysis. Stem Cell Res Ther. 2023;14(1):301. https://doi.org/10.1186/s13287-023-03518-x
13. Nawar AA, et al. Efficacy and safety of stem cell transplantation for multiple sclerosis: a systematic review and meta-analysis of randomized controlled trials. Sci Rep. 2024;14(1):12545. https://doi.org/10.1038/s41598-024-62726-4
14. Elainein MAA, et al. Therapeutic potential of adipose-derived stem cells for diabetic foot ulcers: a systematic review and meta-analysis. Diabetol Metab Syndr. 2025;17(1):9. https://doi.org/10.1186/s13098-024-01523-5
最終更新日:2026.03.19



 Inquiries by Email
Inquiries by Email

