Yes — arterial plaque can be reduced, and atherosclerosis can be partially reversed. But the two tools that do this work on completely different parts of the problem, and it matters which you use for what.
- Statins and PCSK9 inhibitors shrink plaque volume. Modest but real — roughly 1–2% reduction over 18–24 months in imaging trials (ASTEROID, SATURN, GLAGOV). This is the evidence-based way to actually reduce plaque.
- Stem cell therapy does not remove plaque. What it does do is rejuvenate weakened arteries — repair damaged endothelium, calm vascular inflammation, and stimulate collateral vessel growth. That’s an effect statins cannot produce.
Where do those two come together? Stem cell therapy has evidence for improving outcomes after cardiovascular events: heart failure following myocardial infarction (Kavousi 2024, N=1,684: LVEF +3.38, mortality −22%), post-stroke recovery (Xiong 2024 meta-analysis, N=592: mortality OR 0.42, NIHSS −1.63 points, daily function +14.22 points), and critical limb ischemia in ASO (Rigato 2017, N=837: amputation risk −37%, wound healing +59%). Preventing a first MI or stroke in asymptomatic patients? That specific randomized trial doesn’t exist yet — the preventive benefit is biologically plausible from the mechanisms, but hasn’t been directly tested.
This guide explains what each approach — lifestyle, statins, PCSK9 inhibitors, and regenerative medicine — can realistically do, and where stem cell therapy fits alongside (not instead of) conventional cardiovascular care. Written by clinicians, not copywriters. If you came hoping a single injection would erase twenty years of artery damage, this isn’t that article. If you want the honest map, you’re in the right place.
- The Short Answer: What Plaque Reduction Actually Looks Like
- What Atherosclerosis Actually Is (And Why Reduction Is Harder Than Prevention)
- Why Statins Alone Don’t Fully Reverse Atherosclerosis
- How Stem Cell Therapy Rejuvenates Weakened Arteries — Mechanism by Mechanism
- Clinical Evidence for Stem Cell Therapy — Where the Data Actually Exist
- Is There a Cure for Atherosclerosis? A Realistic Answer
- Who Is a Good Candidate for Regenerative Therapy?
- Why Japan? Why CELL GRAND CLINIC?
- Your Treatment Journey: Step by Step
- FAQ
- References
The Short Answer: What Plaque Reduction Actually Looks Like
Different tools do different jobs. Here’s the honest map:
| Approach | What it actually does | Strength of evidence | What it cannot do |
|---|---|---|---|
| Lifestyle (diet, exercise, weight loss) | 0 to −2% plaque volume; lowers CV mortality ~30% | Good — observational + small imaging trials | Repair established endothelial damage on its own |
| High-intensity statins (rosuvastatin 40 mg, atorvastatin 80 mg) | −1% plaque volume over 18–24 months; major event reduction | Strong — ASTEROID, SATURN (RCTs with IVUS) | Stimulate active tissue repair |
| PCSK9 inhibitors on top of statins | Additional −1% plaque volume; 64% of patients show regression | Strong — GLAGOV (RCT, N=968) | Reach the vessel wall biology underneath |
| Regenerative medicine (MSC / ADSC) | Rejuvenates weakened arteries — endothelial repair, anti-inflammation, collateral vessel growth; improves outcomes after events (MI, stroke, ASO) | Moderate — meta-analyses in post-MI HF, post-stroke, CLI | Remove or shrink existing plaque |
So when someone says “plaque is permanent,” they’re half right. Calcified plaque is very hard to reverse. The soft, inflamed, unstable part — the dangerous kind — can shrink with intensive lipid-lowering. And the underlying vessel biology, which statins don’t touch, can at least partially heal with regenerative therapy.
The two approaches aren’t competitors. Statins reduce the LDL flowing past your vessels. Stem cell therapy works on the vessel wall itself. In most clinical situations, you want both.
What Atherosclerosis Actually Is (And Why Reduction Is Harder Than Prevention)
Atherosclerosis isn’t “cholesterol stuck to the wall.” It’s a slow-motion injury to the endothelium — the thin single-cell layer that lines the inside of every artery.
The sequence, simplified:
- Endothelial dysfunction. Hypertension, high LDL, smoking, diabetes, or chronic inflammation damages the endothelium. It starts leaking.
- LDL infiltration. LDL particles slip into the vessel wall and get oxidized.
- Immune response. Macrophages arrive, eat the oxidized LDL, and turn into foam cells.
- Plaque formation. Foam cells plus smooth muscle cells plus debris form a fatty streak, then a fibrous plaque.
- Calcification. Over years, parts of the plaque harden into calcium — this is the part that “can’t be reversed.”
- Rupture risk. If the plaque cap thins and inflammation flares, the plaque can rupture. That’s what causes a heart attack or stroke.
Reduction targets steps 1–4. Reversing step 5 (calcification) is unrealistic with any current therapy. That’s why honest articles about plaque reduction always end up saying “partial.”
The good news: the dangerous plaque — the unstable, inflamed, vulnerable plaque that actually causes events — is not the calcified part. It’s the soft part. And that’s the part therapy can move.
Why Statins Alone Don’t Fully Reverse Atherosclerosis
Statins are a genuine success story. They reduce major cardiovascular events by about 25% per 1 mmol/L LDL reduction, and the evidence is rock-solid. Nothing written below should be read as “don’t take your statin.”
But their mechanism is cholesterol-lowering. They don’t:
- Repair the damaged endothelium directly
- Stimulate new capillary growth in under-perfused tissue
- Reverse the inflammatory phenotype of established foam-cell-rich plaque
- Address the immune dysregulation that keeps plaque inflamed
Imaging trials like ASTEROID (high-dose rosuvastatin, IVUS) and SATURN (rosuvastatin vs atorvastatin) showed about 1% plaque volume reduction over 18–24 months. GLAGOV, which added the PCSK9 inhibitor evolocumab on top of statins, pushed this to around 1% further reduction. These are real, meaningful numbers — but they are modest.
The mechanistic gap is this: statins lower the supply of LDL reaching the vessel wall. They don’t fix the wall itself. If your endothelium is already dysfunctional, reducing the LDL flowing past a leaky wall slows deposits but doesn’t repair the leak.
That’s the space regenerative therapy targets.
How Stem Cell Therapy Rejuvenates Weakened Arteries — Mechanism by Mechanism
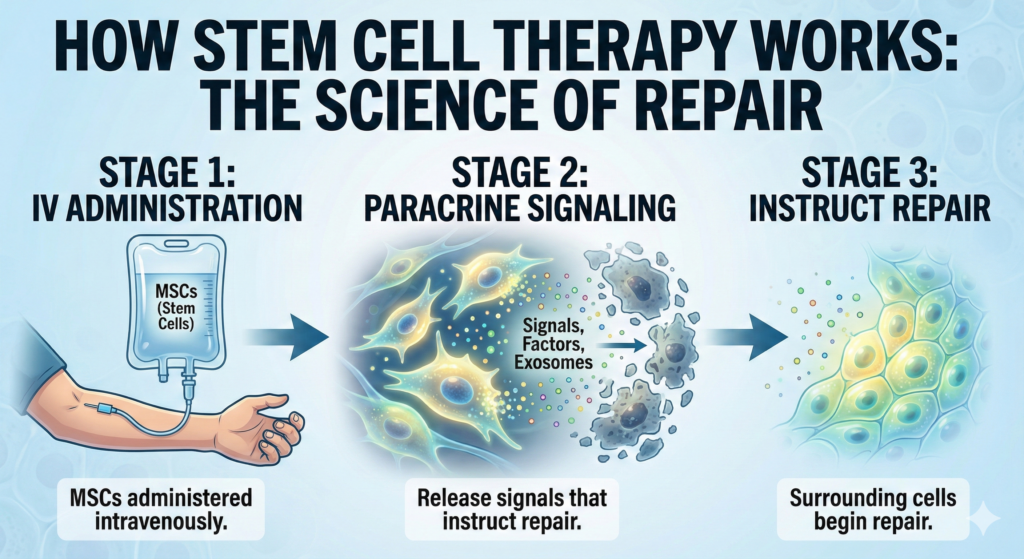
Let’s be clear about what mesenchymal stem cells (MSCs), and specifically adipose-derived stem cells (ADSCs), do and don’t do. They’re not a plaque remover. They don’t dissolve calcium. They’re not stem cells in the “replace every cell” sense most people imagine.
What they are is paracrine signalers — factories that release growth factors, exosomes, and cytokines instructing the patient’s own tissue to repair itself. The target is not the plaque; the target is the vessel wall and its surrounding biology. Three mechanisms matter.
1. Endothelial repair — rebuilding the vessel wall
ADSCs release vascular endothelial growth factor (VEGF), fibroblast growth factor-2 (FGF-2), and hepatocyte growth factor (HGF). These promote endothelial cell proliferation, reduce endothelial apoptosis, and restore nitric oxide production — the signaling molecule that keeps arteries relaxed and anti-inflammatory (Guo et al., Cell Death & Disease, 2020).
In plain language: they help the vessel wall stop leaking. This is the “rejuvenation” part. Healthy endothelium is what a young artery has and an aging, inflamed one has lost.
2. Anti-inflammation — shifting macrophages from “inflame” to “resolve”
The macrophages inside plaque come in two main phenotypes: M1 (pro-inflammatory, plaque-destabilizing) and M2 (anti-inflammatory, tissue-resolving). MSC-derived signals — IL-10, TGF-β, and exosomes carrying specific microRNAs — shift the M1/M2 ratio toward M2.
The clinical meaning: existing plaque becomes less inflamed and less likely to rupture. Think of it as turning a hot, angry plaque into a quiet, stable one. The plaque doesn’t disappear — it becomes safer (Patel et al., International Journal of Molecular Sciences, 2025).
3. Collateral vessel formation — growing new routes around blockages
Through HIF-1α signaling and VEGF release, MSCs stimulate the formation of new small vessels (arteriogenesis and angiogenesis) that bypass partially occluded arteries. This is why MSC therapy has been studied most extensively in post-MI heart failure, post-stroke recovery, and critical limb ischemia — the therapy effectively reroutes blood flow around damaged territory and supplies oxygen to tissue that has been ischemic.
No medication does this. This is where regenerative therapy earns its place alongside statins — not as a plaque remover, but as the biology-repair layer statins cannot provide.
Clinical Evidence for Stem Cell Therapy — Where the Data Actually Exist
Honest answer: stem cell therapy has the strongest evidence for improving outcomes after an ischemic event has already damaged tissue. For preventing a first event in an asymptomatic patient, the direct RCT evidence does not yet exist — the case for prevention rests on the mechanism, not on a prevention trial.
Here are the four evidence buckets, ranked from strongest to most aspirational.
🟢 Bucket 1 — Heart failure after MI (strong evidence)
Kavousi et al., 2024 — meta-analysis of 17 RCTs, 1,684 patients with chronic heart failure (mostly ischemic cardiomyopathy). MSC transplantation:
- Left ventricular ejection fraction +3.38 percentage points (95% CI 1.89–4.87, p<0.001)
- All-cause mortality RR 0.78 (95% CI 0.62–0.99, p=0.04) — a 22% relative reduction
- Hospitalization for HF trended lower overall (RR 0.85, 95% CI 0.71–1.01, p=0.06 borderline); significantly reduced in autologous-cell trials specifically (RR 0.67, p=0.008)
- 6-minute walk distance +37 meters
- Scar size and BNP both reduced
This is a robust signal across multiple RCTs for post-MI / chronic HF patients — the population most at risk of sudden cardiac death from advanced atherosclerosis.
🟢 Bucket 2 — Critical limb ischemia in ASO / PAD (strong evidence, with caveats)
Rigato, Monami & Fadini, 2017 — Circulation Research meta-analysis of 19 RCTs (837 patients), 7 non-RCTs, 41 non-controlled studies (total N ≈ 2,332). Autologous cell therapy in critical limb ischemia:
- Amputation risk reduced by 37%
- Amputation-free survival improved by 18%
- Wound healing improved by 59%
- Ankle-brachial index, transcutaneous oxygen tension, and rest pain all significantly improved
- Intramuscular delivery outperformed intra-arterial
- Effect was larger in diabetic CLI patients
Honest caveat: when the analysis was restricted to placebo-controlled RCTs with low risk of bias, the effect size shrank and significance dropped. This is typical for regenerative therapies — real signals that need more rigorous confirmation — but the trend across study types is consistent enough that many guidelines now consider autologous cell therapy a reasonable option for no-option CLI.
🟢 Bucket 3 — Post-stroke recovery (strong evidence, mortality signal)
Xiong et al., 2024 (Medicine, Baltimore) — meta-analysis of 13 RCTs, 592 ischemic stroke patients, delivered primarily by intravenous infusion on top of standard care (with intra-arterial delivery used in a minority of studies; the paper’s own discussion notes the optimal delivery route remains to be determined). Stem cell therapy produced:
- NIHSS neurological severity −1.63 points (95% CI −2.69 to −0.57, p=0.003) — improvements comparable to weeks of intensive rehabilitation
- Barthel Index daily function +14.22 points (95% CI 3.95–24.48, p=0.007) — a meaningful shift in the ability to perform activities of daily living
- All-cause mortality OR 0.42 (95% CI 0.23–0.79, p=0.007) — in plain terms, if 100 patients in the control group died, fewer than half that number died in the stem cell group
- Modified Rankin Scale trended favorably (MD −0.32, p=0.05 — borderline)
This is not a modest trend. A mortality halving on top of standard stroke care — the most fundamental outcome — is a clinically meaningful signal in the population most at risk of dying from advanced cerebrovascular atherosclerosis.
Safety context (AMASCIS trial — de Celis-Ruiz et al., 2022) — small phase II RCT (13 treated). 24-month safety confirmed with no injection-related AEs and no tumor development. Efficacy signals were in the right direction but not statistically significant at this sample size — a reminder that small single-center studies often can’t detect the effect the larger meta-analysis finds.
Honest caveats: Xiong 2024 notes heterogeneity across cell sources and urges larger confirmatory RCTs. But the direction and magnitude of benefit are consistent enough across 13 RCTs that this is the strongest mortality signal in the regenerative cardiovascular literature outside Kavousi.
🟡 Bucket 4 — Primary prevention of MI or stroke (plausible, not directly proven)
No randomized trial has taken asymptomatic patients with early atherosclerosis, given them MSC therapy, and followed them to see whether future MI or stroke incidence dropped. That study doesn’t exist yet.
What we can say honestly:
- The biological mechanisms documented above (endothelial repair, anti-inflammation, collateral formation) are exactly the mechanisms that would, in theory, reduce first-event risk
- The downstream “post-event” evidence from Buckets 1–3 is consistent with the mechanism working in humans
- A preventive benefit is biologically plausible, but not yet directly demonstrated
This is the most honest framing for any patient considering regenerative therapy for cardiovascular prevention. The ceiling of what we know is different from the ceiling of what is probably true, and both matter when making a clinical decision.
What these numbers don’t mean
- They don’t mean stem cells cure atherosclerosis
- They don’t mean every patient responds
- They don’t mean the effect is permanent (some trials show benefits fading by 24 months without boosters)
- They don’t mean this is first-line therapy — statins and lifestyle remain first-line for almost every patient
Is There a Cure for Atherosclerosis? A Realistic Answer
“Cure” is the wrong word. Here’s a more useful framing.
Atherosclerosis is a lifelong biology, driven by genes, diet, blood pressure, inflammation, and time. You can’t cure time. What you can do, by combining the right tools, is:
- Slow progression to near-zero with aggressive statins + lifestyle (well-established in trials)
- Shrink soft plaque volume by roughly 1–2% over 18–24 months with high-intensity statins ± PCSK9i (ASTEROID, SATURN, GLAGOV)
- Stabilize vulnerable plaque so it doesn’t rupture (statins + anti-inflammatory mechanisms)
- Repair endothelial function in the segments that are still repair-capable (regenerative therapy’s domain)
- Grow collateral vessels around existing blockages (regenerative therapy’s domain)
- Improve outcomes after a cardiovascular event has occurred (regenerative therapy has the strongest evidence here)
That’s not a cure. It’s partial reversal plus stabilization plus vessel rejuvenation. For most patients, that’s the difference between a stroke at 62 and a stroke at 82 — or no stroke at all.
Regenerative medicine doesn’t replace statins, and it isn’t a substitute for lifestyle work. It adds a layer that statins and lifestyle alone can’t provide: active tissue repair rather than supply reduction.
Who Is a Good Candidate for Regenerative Therapy?
Reasonable candidates share several features:
- Established atherosclerosis on imaging (carotid plaque, coronary calcium, or CCTA findings) but not critical stenosis requiring urgent intervention
- On optimized medical therapy (appropriate statin, blood pressure control, diabetes management) but wanting to go further
- No active malignancy
- No severe uncontrolled autoimmune disease
- Age roughly 40–80 — biological age matters more than chronological age
- Committed to lifestyle work (we don’t accept candidates who see regenerative therapy as a shortcut)
We don’t recommend regenerative therapy for patients with acute coronary syndrome, active stroke, or severe critical-limb ischemia requiring urgent care. Those are surgical or interventional situations first.
Why Japan? Why CELL GRAND CLINIC?
Japan’s Regulatory Standard
Japan is one of the few countries in the world with a dedicated national law governing regenerative medicine. Under the Act on the Safety of Regenerative Medicine (2014), every stem cell treatment plan must be reviewed by an independent MHLW-Certified Special Committee before it can be offered to patients. All outcomes are reported to a national safety database. This is not self-regulation — it is government-enforced quality assurance at the level of pharmaceutical manufacturing. Mexico, Thailand, and many other “stem cell tourism” destinations operate with no equivalent oversight.
| Factor | Japan (MHLW-certified) | Mexico | Thailand |
|---|---|---|---|
| Government Oversight | MHLW certified ✓ | Limited regulation | Emerging regulation |
| Cell Quality Standards | CPC-grade, 7-week culture ✓ | Varies by clinic | Varies by clinic |
| Cell Count per Treatment | Up to 200 million ✓ | Typically varies | Typically varies |
| Autologous (Own Cells) | Yes ✓ | Often allogeneic | Mixed |
| Physician Credentials | NIH-trained MD ✓ | Varies | Varies |
| National Safety Database Reporting | Mandatory ✓ | Not required | Not required |
Comparison based on general regulatory frameworks. Japan’s MHLW certification system (Act on the Safety of Regenerative Medicine) provides government-level oversight unique among major stem cell therapy markets.
Cell Grand Clinic: Academic-Grade Quality at the Point of Care
Cell Grand Clinic in Shinsaibashi, Osaka holds 10 Type II Government Licenses — the highest outpatient classification under Japan’s regenerative medicine regulatory system. The stem cells are cultured at a government-certified Cell Processing Center (CPC, License: FA5250001) meeting GMP-equivalent pharmaceutical manufacturing standards. Every batch undergoes multi-marker purity and potency testing before release. The culture period is approximately 7 weeks, during which cells are expanded to up to 200 million — a cell count that substantially exceeds what many international clinics offer.

Dr. Yuichi Wakabayashi, M.D., Ph.D., Medical Director, brings a combination of credentials rare in regenerative medicine: NIH Research Fellow (neurological disorders), Diplomate of the American Board of Regenerative Medicine (ABRM), first author of a world-first PET tracer study in collaboration with Pfizer, published in the Journal of Nuclear Medicine, and featured in The Wall Street Journal as a “Next Era Leader.” Dr. Wakabayashi communicates directly in English — no interpreters.
Your Treatment Journey: Step by Step
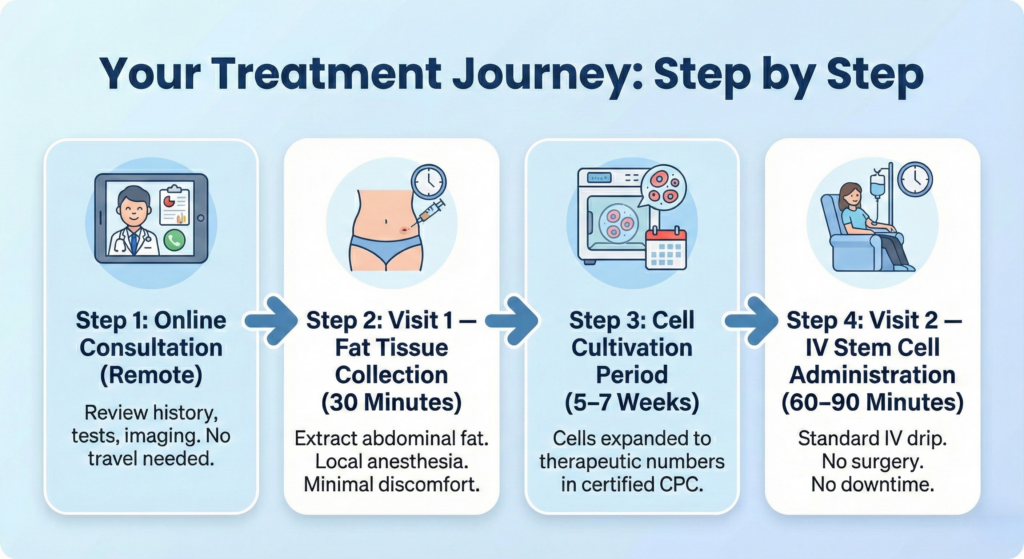
Step 1: Online Consultation (Remote)
We review your medical history, blood tests, and imaging (MRI/CT) remotely. No travel required for this initial assessment. Dr. Wakabayashi personally evaluates every case.
Step 2: Visit 1 — Fat Tissue Collection (30 Minutes)
Under local anesthesia, we extract a small amount of fat tissue from your abdomen. This is the source of your autologous stem cells. The procedure involves minimal discomfort and requires no general anesthesia.
Step 3: Cell Cultivation Period (7 Weeks)
You return home while your stem cells are expanded to therapeutic numbers—100 million or 200 million cells—in our government-certified CPC under pharmaceutical-grade conditions.
Step 4: Visit 2 — IV Stem Cell Administration (60–90 Minutes)
Your stem cells are administered via a standard intravenous drip. No surgery, no general anesthesia, no downtime. You can return to your hotel or resume light activities immediately after the session.
If you have other age-related disease, check below.
▶Stem cell therapy for diabetes
▶Stem cell therapy for chronic pain
FAQ
Can atherosclerosis be reversed with diet alone?
Diet alone can produce small plaque reductions — roughly 1% volume reduction over a year in the strictest trials (like the Ornish and Esselstyn programs). That’s real but modest. For most patients, diet is a necessary foundation, not a sufficient treatment.
Can atherosclerosis be reversed with exercise?
Exercise mainly improves endothelial function and collateral circulation rather than directly shrinking plaque. It lowers cardiovascular mortality powerfully (around 30% in observational data) — but its plaque-volume effect is smaller than statins. Both together are much more effective than either alone.
Can statins reverse atherosclerosis?
Partially. High-intensity statins (rosuvastatin 40 mg, atorvastatin 80 mg) produce about 1% plaque volume reduction over 18–24 months in imaging trials (ASTEROID, SATURN). Adding PCSK9 inhibitors on top can push this further. Statins are the first and most important step for almost every patient with atherosclerosis.
How long does it take to reduce arterial plaque?
Measurable plaque volume changes typically appear between 6 and 18 months on serial imaging. Inflammation markers (hs-CRP) and endothelial function change much faster, usually within weeks.
Can stem cells remove arterial plaque?
No — and any clinic telling you otherwise is overselling. Stem cells (specifically MSCs and ADSCs) do not dissolve plaque, do not shrink calcified lesions, and do not work like surgery or atherectomy. What they actually do is target the underlying vessel biology: endothelial repair, macrophage phenotype shift (M1 to M2), and collateral vessel growth. The clinical payoff shows up not as a smaller plaque on imaging, but as improved outcomes after cardiovascular events — better ejection fraction after MI (Kavousi 2024), lower amputation rates in CLI (Rigato 2017), and roughly halved post-stroke mortality (Xiong 2024, OR 0.42). Plaque reduction is the statin’s job. Vessel rejuvenation is the stem cell’s job.
Is stem cell therapy for atherosclerosis FDA-approved in the US?
No. As of 2026, no stem cell therapy has full FDA approval for atherosclerosis or coronary artery disease. Several trials are ongoing. In Japan, under the Act on the Safety of Regenerative Medicine, autologous ADSC therapy is permitted at licensed clinics (including Cell Grand Clinic) under MHLW-approved protocols.
How much plaque reduction is realistic?
For plaque volume, the honest target is roughly 1% over 18–24 months with high-intensity statins (ASTEROID −0.98% at 24 months; SATURN −0.99% to −1.22% at 104 weeks), and about another 1% on top of that when a PCSK9 inhibitor is added (GLAGOV evolocumab −0.95% vs placebo +0.05% at 76 weeks). So the practical ceiling on combination therapy is around 1–2% total volume reduction across the first two years — modest but meaningful when maintained. Lifestyle adds a bit more. Regenerative therapy does not add to these plaque-volume numbers — its benefit is on the vessel biology, not the plaque itself. For soft, inflamed plaque specifically, the reduction can be somewhat higher than for mixed plaque; for calcified plaque, changes are minimal.
Can you unclog arteries without surgery?
Partially, yes — but it’s important to match the tool to the job. Diet and high-intensity statins (with PCSK9i where indicated) can reduce soft plaque volume and stabilize vulnerable plaque. Regenerative therapy does not unclog arteries, but it does grow collateral vessels that can bypass existing blockages and improve tissue oxygenation — which is why it has evidence in post-MI heart failure and critical limb ischemia. Once an artery reaches critical stenosis (typically 70%+ blockage causing symptoms), surgical or catheter-based intervention is usually required. Regenerative therapy works best as a complement to optimized medical therapy, not as a rescue for severe stenosis.
References
- Kavousi S, Hosseinpour A, Bahmanzadegan Jahromi F, Attar A. Efficacy of mesenchymal stem cell transplantation on major adverse cardiovascular events and cardiac function indices in patients with chronic heart failure: a meta-analysis of randomized controlled trials. J Transl Med. 2024;22:786. DOI: 10.1186/s12967-024-05352-y
- Patel T, Mešić J, Meretzki S, et al. Therapeutic potential and mechanisms of mesenchymal stem cells in coronary artery disease: narrative review. Int J Mol Sci. 2025;26(11):5414. DOI: 10.3390/ijms26115414
- Guo Y, Yu Y, Hu S, Chen Y, Shen Z. The therapeutic potential of mesenchymal stem cells for cardiovascular diseases. Cell Death Dis. 2020;11:349. DOI: 10.1038/s41419-020-2542-9
- de Celis-Ruiz E, Fuentes B, Alonso de Leciñana M, et al. Final results of allogeneic adipose tissue-derived mesenchymal stem cells in acute ischemic stroke (AMASCIS): a phase II, randomized, double-blind, placebo-controlled, single-center, pilot clinical trial. Cell Transplant. 2022;31:09636897221083863. DOI: 10.1177/09636897221083863
- Nissen SE, Nicholls SJ, Sipahi I, et al. Effect of very high-intensity statin therapy on regression of coronary atherosclerosis: the ASTEROID trial. JAMA. 2006;295(13):1556–1565.
- Nicholls SJ, Ballantyne CM, Barter PJ, et al. Effect of two intensive statin regimens on progression of coronary disease. N Engl J Med. 2011;365:2078–2087. (SATURN trial)
- Nicholls SJ, Puri R, Anderson T, et al. Effect of evolocumab on progression of coronary disease in statin-treated patients: the GLAGOV randomized clinical trial. JAMA. 2016;316(22):2373–2384.
- Rigato M, Monami M, Fadini GP. Autologous cell therapy for peripheral arterial disease: systematic review and meta-analysis of randomized, nonrandomized, and noncontrolled studies. Circ Res. 2017;120(8):1326–1340. DOI: 10.1161/CIRCRESAHA.116.309045
- Xiong Y, Guo X, Gao W, et al. Efficacy and safety of stem cells in the treatment of ischemic stroke: a meta-analysis. Medicine (Baltimore). 2024;103(12):e37414. DOI: 10.1097/MD.0000000000037414
最終更新日:2026.04.23



 Inquiries by Email
Inquiries by Email

