Stem cell therapy for a torn meniscus is a non-surgical injection treatment that uses mesenchymal stem cells (MSCs) — typically harvested from your own body fat — to reduce knee inflammation, support the joint’s natural repair environment, and relieve the pain and stiffness of a meniscus tear without removing any tissue. At Cell Grand Clinic in Osaka, Japan, the treatment costs approximately $15,000 to $30,000 USD per session and is delivered under Japan’s strict Ministry of Health, Labour and Welfare (MHLW) regulatory framework. The strongest published human evidence is a 2023 Phase I clinical trial showing safety and functional improvement at 12 months. Stem cell therapy does not physically glue a torn meniscus back together, but it can meaningfully reduce pain and restore range of motion — and for many patients, it eliminates the need for surgery.
If You’re Reading This, You Probably Don’t Want Surgery. Here’s the Real Story.- Why No Meniscus Treatment Is Perfect ?
- Why Meniscus Tears Are So Hard to Heal?
- What the Clinical Evidence Actually Shows
- The Honest MRI Answer: Your Meniscus Won’t Look “Brand New” — And That’s OK
- How Stem Cell Works
- Stem Cells vs. Surgery vs. Physical Therapy: The Fair Comparison
- Stem Cell Therapy for Torn Meniscus Cost: What You’ll Actually Pay
- Why Cell Grand Clinic? The “Grand Stem Cell” Promise
- Treatment Process for International Patients
- Who Is a Good Candidate?
- Frequently Asked Questions
- The Bottom Line
- References
If You’re Reading This, You Probably Don’t Want Surgery. Here’s the Real Story.
Your knee hurts. Your MRI says “meniscus tear.” Your orthopedic surgeon has offered to “clean it up” with arthroscopic surgery. But something is telling you to slow down and look at your options.
You’re in good company. Every week we talk to people from all over the world who are in exactly this spot — and most of them have the same three questions:
- Can stem cell therapy really help my meniscus, or is it just hype?
- How much does it cost, and what am I actually paying for?
- Is it a better choice than surgery — really?
This article answers all three in plain English, with no oversell and no pretending the evidence is bigger than it is. We’ll explain what a 2023 clinical trial actually found, show you the honest cost range, and — most importantly — walk through something most clinics won’t admit: no option for a torn meniscus is perfect. Not surgery. Not physical therapy. And not stem cells either. Choosing well means understanding the trade-offs, not swallowing the marketing.
Why No Meniscus Treatment Is Perfect ?
Most patients assume meniscus surgery is a near-certain fix. It isn’t. Here’s what the research actually shows, in plain language.
Arthroscopic surgery comes in two flavors, and both have real downsides.
- Partial meniscectomy (cutting out the torn portion) is the more common operation. It usually relieves pain fast, but every piece of meniscus removed increases your risk of knee osteoarthritis down the road. Long-term studies have been warning about this for over a decade — the meniscus you lose today is meniscus you’ll miss in ten years.
- Meniscus repair (stitching the torn piece) tries to save the tissue. Great in theory — but the published data is sobering. A 2022 meta-analysis of 41 studies covering over 1,900 patients found an overall meniscus repair failure rate of about 12% across all techniques [9]. In higher-risk groups — younger, more active patients, and certain repair techniques — failure rates climb substantially higher, with some recent series reporting around 23% re-tear in athletes undergoing all-inside repair [10]. Success also depends on where the tear is, how old it is, and the patient’s age.
In other words: even the “best case” surgery can leave a meaningful percentage of patients back in the operating room — with the risk rising sharply in younger, more demanding knees.
“Just do physical therapy” isn’t a free lunch either. Conservative care works for some people, but it mainly manages symptoms without changing the underlying tear or inflammation. For many patients, it means living with chronic pain and hoping it plateaus.
So what about stem cell therapy? It has real limits too (we’ll get to those). But unlike the other options, it targets the biology of the injured knee — the inflammation, the irritation, the destructive chemistry around the tear — without cutting anything away. For the right patient, that’s a genuinely different kind of option, not just “another injection.”thing away. For the right patient, that’s a genuinely different kind of option, not just “another injection.”
Why Meniscus Tears Are So Hard to Heal?
The meniscus is a tough, C-shaped fibrocartilage cushion between your thigh bone (femur) and shin bone (tibia). It absorbs shock, spreads out your weight, and stabilizes the knee. It’s doing a lot of work. But here’s the catch: most of it has almost no blood supply.
Anatomists describe three vascular zones — think of them like rings on a target:
- Red-red zone (outer 1/3): Good blood flow. Tears here sometimes heal with conservative care or surgical repair.
- Red-white zone (middle 1/3): Marginal blood flow. Healing is unpredictable.
- White-white zone (inner 2/3): Essentially no blood flow. Tears here almost never heal without outside help.
Your body can heal most things — bruises, cuts, even broken bones — because blood delivers oxygen, nutrients, and repair signals. But blood can’t reach most of the meniscus. A 2025 review in Journal of Clinical Medicine confirmed that the vascular pattern of a torn meniscus largely determines its healing potential [8].
For decades, surgeons responded by simply cutting the damaged part out. We now know that trade-off came with a long-term price: losing meniscus means losing cartilage, which means earlier osteoarthritis. Stem cell therapy tries to do something different — deliver the repair signals the meniscus can’t get from your bloodstream, directly into the joint.
What the Clinical Evidence Actually Shows
We’re not going to hide behind jargon. Here’s what the research — and our own patients — actually show.
The Strongest Clinical Trial So Far
In 2023, a team published a clinical trial in Signal Transduction and Targeted Therapy that tested something quite direct: take 18 patients with meniscus injuries, inject regenerative cells into their knees, and see what happens over 12 months. The results were encouraging on three fronts:
- Safety. Nothing alarming showed up over a full year of follow-up.
- MRI imaging. Measurable improvement in the meniscus area was observed.
- Function. Patient knee scores — pain, mobility, daily activity — improved meaningfully.
This is the strongest human evidence we have specifically for cell therapy in meniscus injury. One important nuance: the cells used in that trial were a research product called IMRCs (Immunity-and-Matrix-Regulatory Cells), which are different from the autologous adipose-derived mesenchymal stem cells Cell Grand Clinic uses. But the trial still matters, because it’s the first modern peer-reviewed human study to show that delivering living, quality-controlled cells into a knee with a meniscus injury is safe and biologically active over 12 months. It’s a proof-of-principle for the whole approach, not a 1,000-patient mega-trial.
The Surgical World Is Already Adding Biology to Meniscus Repair
Here’s a shift that surprises most patients. In 2025, an international survey by the Meniscus International Network (MenIN) Study Group — 42 experienced knee surgeons from around the world — found that about 90% of them now routinely add some form of biologic or mechanical augmentation to their meniscus repairs. The most common techniques are mechanical (rasping, trephination, marrow venting) to stimulate the body’s own healing response; about one-third of surveyed surgeons also add platelet-rich plasma (PRP) or bone marrow aspirate concentrate (BMAC) in specific cases. That’s a dramatic change from 20 years ago, when meniscus surgery was purely mechanical — stitch the tear, close up, done. The mainstream surgical world increasingly agrees: pure mechanics alone isn’t enough; the knee needs biological help to heal.
Strong Adjacent Evidence from Knee Osteoarthritis
Meniscus damage and knee osteoarthritis share the same underlying biology, and the research on stem cells for knee osteoarthritis is more mature. A 2025 systematic review and meta-analysis pooled 8 randomized trials covering 502 patients and found that intra-articular mesenchymal stem cell injection significantly reduced pain and improved knee function, with a side effect profile similar to placebo. Subgroup analysis showed that high-dose MSC injections outperformed low-dose, and a separate large Phase III randomized, double-blind, placebo-controlled trial of 261 patients used exactly the approach Cell Grand Clinic uses — autologous adipose-derived mesenchymal stem cells at 100 million cells per injection — and confirmed the same pattern of durable pain and function improvement at 6 months.
Summary of the Evidence (Honest Version)
| Claim | Evidence Type | Verdict |
|---|---|---|
| Safe for intra-articular use | Phase I meniscus trial + multiple knee OA trials | Strong evidence ✓ |
| Reduces pain & improves function | Phase I trial + BMAC case series + OA meta-analysis | Consistent signal ✓ |
| Physically “repairs” a tear like new | MRI stabilization, not true reattachment | Not really — explained below |
| High-dose adipose-derived MSCs outperform low-dose | 2025 meta-analysis subgroup analysis + 261-patient Phase III RCT using 100M cells | Supported ✓ |
| Proven by a 1,000-patient RCT specifically for meniscus | None yet | Not yet — anyone claiming otherwise is oversimplifying |
Summarized from peer-reviewed sources indexed on PubMed — see References at the end of this article.
Send it to us and we’ll tell you honestly whether stem cell therapy is worth considering for your specific tear. WhatsApp and email are free, no obligation.
The Honest MRI Answer: Your Meniscus Won’t Look “Brand New” — And That’s OK
This is the most important section in this article, so we’re going to be very direct.
Stem cell therapy does not physically glue a torn meniscus back together. Anyone who shows you a before/after MRI where a tear “disappears” and the meniscus looks new after one injection is either misrepresenting the science or showing you an unrepresentative case.
So what does happen on imaging? In Dr. Wakabayashi’s clinical experience — treating meniscus patients at Cell Grand Clinic and reviewing their follow-up scans — the MRI pattern typically shifts in a specific and interesting way:
What we usually see: an acute-looking, fluid-bright, angry tear signal calms down and begins to look more like a quiet, chronic, degenerative-level change. The tear is often still visible. But the inflammation around it, the irritation in the joint, and the destructive chemistry — those calm down considerably.
In plain English: your MRI doesn’t become normal. It becomes quiet. And a quiet knee is what lets you walk, sleep, and move without pain.
And here’s the part that matters most: even though the MRI still shows “something,” the symptoms often get dramatically better. Pain with walking and stairs eases up. Stiffness loosens. Catching and locking sensations calm down. Range of motion comes back. We have one model patient in particular whose case beautifully illustrates this — the tear is still visible on the post-treatment scan, but the acute fire has gone out, and clinically he is a completely different person than before treatment. (We can share case details, with permission, during a private consultation.)
If a clinic promises your MRI will look “completely normal” after one shot, that’s a warning sign about the clinic — not a reason to believe it. Honest expectations protect you.
How Stem Cell Works
If stem cells don’t magically become new meniscus, what are they doing in there? Four things, all at once:
- They turn down inflammation. A torn meniscus triggers a chronic inflammatory environment in the knee — synovitis, effusion, and destructive cytokines. MSCs release anti-inflammatory mediators (IL-10, TGF-β, IL-1 receptor antagonist) that break this cycle. It’s like a thermostat for your knee.
- They help your body repair what it can. Stem cells don’t usually become new tissue themselves. Instead, they create a friendly environment for your body’s own healing machinery to work — what researchers call a “permissive niche” for repair.
- They calm down your immune system. An irritated knee is partly an immune problem. Stem cells reprogram overactive T cells, B cells, and macrophages back toward normal through paracrine signaling.
- They give you relief that lasts. A cortisone shot wears off in weeks. Hyaluronic acid lubricates but doesn’t fix anything. Stem cells address the underlying biology, which is why benefit tends to build over months rather than fade.
Stem Cells vs. Surgery vs. Physical Therapy: The Fair Comparison
Here’s the comparison most clinic websites won’t give you — because it doesn’t pretend any option is flawless.
| Question | Stem Cell Therapy | Arthroscopic Surgery | Physical Therapy Only |
|---|---|---|---|
| How invasive? | Injection, 30 min | Operating room, anesthesia | Not invasive |
| When can I walk? | Same day | Days to weeks | No downtime |
| Preserves meniscus? | Yes | Repair: tries to. Meniscectomy: removes tissue. | Yes |
| Long-term OA risk? | Not increased | Meniscectomy raises it | May progress untreated |
| What can go wrong? | Temporary soreness. Not everyone responds. | 12–25% failure rate (varies by tear & technique). Infection, DVT, anesthesia risk. | Pain may persist. Tear unchanged. |
| Is it “perfect”? | No — but works well for many tears | No — especially for degenerative tears | No — often manages, doesn’t fix |
No single option is perfect. The right choice depends on your tear pattern, your age, your activity goals, and your tolerance for surgery vs. injection. A good clinic will tell you when surgery is actually the better answer.
A straight answer about when we recommend surgery instead. If you have a young, mechanically unstable, bucket-handle tear that’s locking your knee, surgery is often the right call — and we’ll tell you so. Stem cell therapy is not a universal replacement for the operating room. It’s a real alternative for degenerative tears, horizontal tears, complex tears, and most meniscus injuries in adults over 40 who want to keep their meniscus and avoid the long-term consequences of cutting it out.
If you also have advanced knee osteoarthritis along with your meniscus tear, you may want to read our companion guide on knee stem cell therapy for osteoarthritis.
Stem Cell Therapy for Torn Meniscus Cost: What You’ll Actually Pay
Let’s talk money, because this is usually the next question.
When patients research stem cell therapy for torn meniscus cost, they find prices all over the map — from suspiciously cheap “stem cell injections” at $2,000 in wellness spas, to premium, regulated treatments at $15,000–$30,000 at certified clinics. That’s a huge spread, and there’s a reason for it.
The real cost of a premium stem cell treatment in Japan — including Cell Grand Clinic — typically lands between $15,000 and $30,000 USD per session for a single knee. Here’s what drives that price.
| What You’re Paying For | Why It Matters |
|---|---|
| A full 100 million cells | The 2025 meta-analysis showed lower doses just don’t work as well. Under-dosing is the easiest way for a clinic to cut costs while keeping the word “stem cells” on the brochure. |
| A 4-7-week GMP cell culture | Rushed 2-week cultures save money but produce weaker cells. The extra culture time is what delivers strong, viable, quality-tested cells. |
| Japanese government oversight | Japan’s MHLW regulatory framework requires approved treatment plans, ethics review, and continuous monitoring — none of which is free, but all of which protects you. |
| ISCT-standard quality verification | Identity testing (CD73/CD90/CD105) and viability verification (≥95%) is laboratory work that costs real money. Clinics that skip this don’t really know what they’re injecting. |
| Your own cells (autologous) | Cell Grand Clinic cultures cells from your own fat. More expensive than off-the-shelf donor cells, but no risk of immune rejection and no mystery about the source. |
| Bridge pain management | PRP or exosome therapy during Visit 1 to keep you comfortable during the 7-week culture wait. Some clinics quote this separately. |
A detailed, personalized quote is provided during the initial remote consultation — before you book any travel.
Why the $2,000 “stem cell shot” isn’t really the same thing. Those ultra-cheap offers almost never deliver 100 million quality-verified cells. They often contain stromal vascular fraction (unprocessed fat cells), allogeneic cell products of unknown origin, or cells cultured without quality controls. The price gap isn’t a rip-off — it reflects a real difference in what’s being injected into your knee.
Send us your MRI and we’ll respond within 48 hours with a realistic assessment. No credit card, no booking commitment.
Why Cell Grand Clinic? The “Grand Stem Cell” Promise
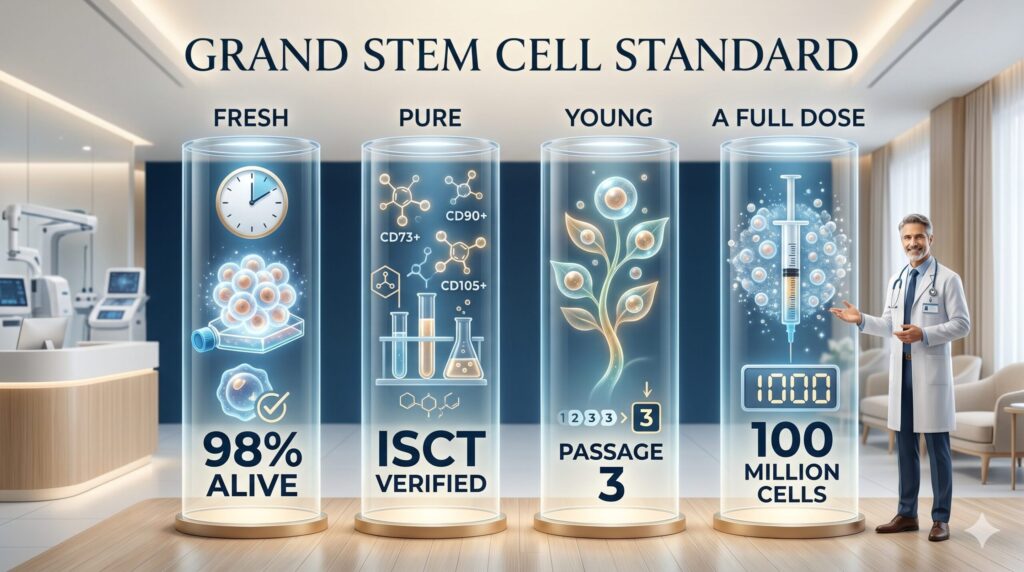
“Stem cell therapy” is one of those phrases that sounds the same everywhere but can mean very different things. So we built our own quality standard to make it clear — and we put it on a certificate we hand every patient.
We call it the Grand Stem Cell standard. Cells only qualify if they pass all four of these tests:
1. Fresh. We culture your cells specifically for you, starting only after you confirm your treatment. No pre-made cells sitting in a freezer. We verify almost 98% of the cells are alive before we inject anything — well above the industry average of 70–85%. Dead cells don’t heal anything.
2. Pure. Every batch is tested against International Society for Cell & Gene Therapy (ISCT) standards — approximately 99% CD73+, CD90+, CD105+ expression, with contaminants (CD45−, CD34−) eliminated. This sounds like a given, but plenty of clinics skip this step.
3. Young. Stem cells age every time you culture them, and old cells lose their regenerative power. Cell Grand Clinic caps cultures at Passage 3 so the cells you receive are still biologically vigorous.
4. A full dose. Every patient receives a guaranteed 100 million cells — the amount the 2025 meta-analysis identified as optimal. No under-dosing.
Every patient also receives a Certificate of Quality with lab verification data, so you know exactly what you got.
Japanese government oversight. Japan’s Act on the Safety of Regenerative Medicine (2014) is one of the world’s strictest legal frameworks for stem cell treatment. Clinics must submit detailed treatment plans to MHLW-certified committees for approval before treating any patient. Cell Grand Clinic holds 13 approved treatment plans — one of the broadest portfolios of any regenerative medicine clinic in Japan — and operates under continuous government monitoring.
Expert leadership. Every treatment is personally overseen by Dr. Yuichi Wakabayashi, Diplomate of the American Board of Regenerative Medicine (ABRM), a former NIH Research Fellow, and featured in The Wall Street Journal. Cell culture protocols are developed in collaboration with Professor Takahiro Ochiya’s research team, a globally recognized group in exosome and stem cell biology.
▶ Learn more about Cell Grand Clinic
▶ See our knee osteoarthritis stem cell page
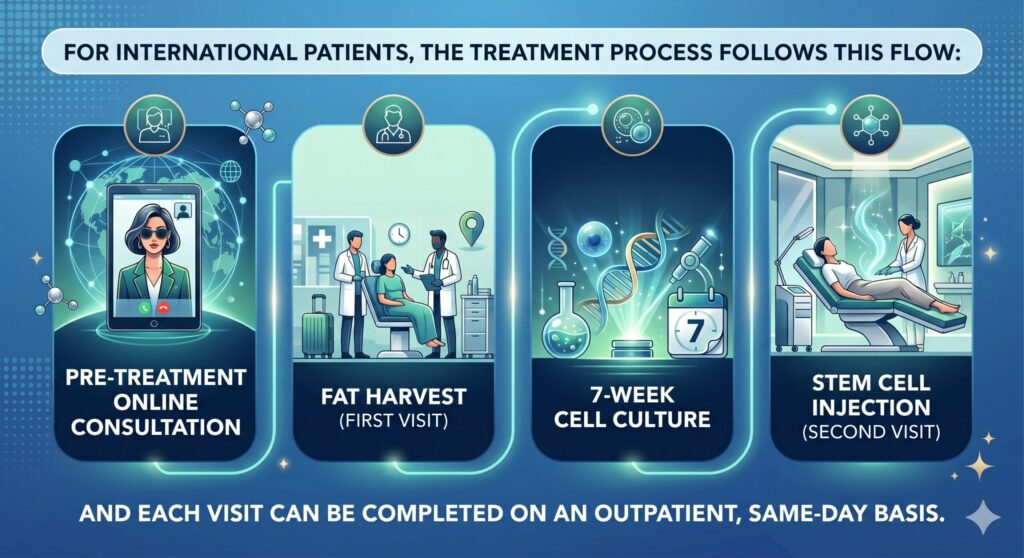
Treatment Process for International Patients
Step 1 — Remote Consultation. Send us your MRI and exam notes. We review everything and tell you honestly whether you’re a good candidate before you book any flights.
Step 2 — Visit 1 in Osaka (Day 1). A quick outpatient fat harvest — about 30 minutes, under local anesthesia. You walk out the same day. Most international patients turn this into a short Osaka or Kyoto trip.
Don’t want to wait 7 weeks in pain? During Visit 1, your doctor can also give you PRP or exosome therapy directly into the knee to manage symptoms while your stem cells are being cultured.
Step 3 — Cell Culture (7 Weeks). You fly home. Meanwhile, your cells are multiplied in a GMP-certified Cell Processing Center until you have 100 million Grand Stem Cells ready to go.
Step 4 — Visit 2 in Osaka (Week 8). Come back for the injection. The appointment is about 30 minutes. You walk out the same day and can fly home within a day or two.
Who Is a Good Candidate?
We review every case individually. If it’s not right for you, we’ll tell you.
Frequently Asked Questions
How much does stem cell therapy for a torn meniscus cost?
A properly regulated stem cell treatment in Japan typically runs $15,000 to $30,000 USD per session for one knee. That includes 30-100 million of your own cells, cultured over 4-7 weeks, quality-tested, and delivered under Japanese government oversight. If you see quotes dramatically cheaper than that, it’s usually because the product is fundamentally different — lower cell counts, uncultured fat cells, or donor cells from an unknown source. Cell Grand Clinic provides an itemized quote after the free remote review, so there are no surprises.
Can stem cell therapy really “repair” a torn meniscus?
Honest answer: not in the Hollywood sense. It won’t make your MRI look brand new or glue the torn fibers back together. What it does do — and what the 2023 Phase I clinical trial [1] confirmed — is calm the inflammation around the tear, stabilize the injury, and shift the MRI from an angry acute-looking tear toward a quieter, chronic-looking pattern. That shift comes with real, meaningful improvements in pain and range of motion.
Is there actually clinical evidence for this, or is it just marketing?
Real evidence exists. The strongest human data specific to meniscus injury is a 2023 Phase I dose-escalation trial of 18 patients who received intra-articular cell injection — safety was confirmed over 12 months with improved MRI and functional knee scores [1]. (One note: that trial used a research cell product called IMRCs, which is different from the autologous adipose-derived stem cells Cell Grand Clinic uses — but the safety and biological-activity signal is directly relevant.) Separately, case series of meniscus repairs augmented with bone marrow concentrate show large improvements [3], with one long-term series sustained to 10 years [4]. The adjacent research on stem cells for knee osteoarthritis is more mature: a 2025 meta-analysis of 8 randomized trials covering 502 patients [5] and a 261-patient Phase III RCT of autologous adipose-derived MSCs at 100 million cells per injection [6] both show consistent pain relief and function improvement. Is there a 1,000-patient meniscus-specific RCT? No, not yet. But is there a real, reproducible signal across multiple human studies? Yes.
How is stem cell therapy different from PRP?
PRP (platelet-rich plasma) is made from your blood and delivers growth factors. It can reduce inflammation and often helps, but it doesn’t contain stem cells. Stem cell therapy delivers actual living, quality-verified mesenchymal stem cells that do more complex things — they signal to your immune system, support repair, and have longer-lasting effects. At Cell Grand Clinic, we often use PRP or exosome therapy as a “bridge” to keep patients comfortable during the 7-week cell culture — but it’s not a substitute for stem cell therapy.
Will I definitely avoid surgery?
Many patients do — but we can’t promise it, and no honest doctor will. In our experience, a significant proportion of patients with degenerative or complex tears avoid or postpone surgery after stem cell treatment and return to normal activity. Some patients, especially those with mechanically unstable bucket-handle tears, will still need surgery. We assess every case individually before we recommend treatment.
How soon will I feel better?
Most patients start noticing improvement in 1 to 3 months, with benefit typically continuing to build over 6 to 12 months as inflammation settles. It’s not an overnight cortisone-style fix — it’s more like a slow, steady turning-down of the noise in your knee. The 2023 Phase I trial [1] followed patients for 12 months with sustained functional improvement.
Is it safe?
Yes, based on both published evidence and clinical experience. The 2023 Phase I trial reported no serious issues over 12 months of follow-up [1]. The 2025 meta-analysis of 502 knee osteoarthritis patients [5] found that stem cell injections had a side effect profile similar to placebo. Because Cell Grand Clinic uses your own (autologous) cells, immune rejection is not a concern. The most common side effects are temporary soreness or swelling at the injection site for a few days.
Can I walk right after treatment?
Yes. Unlike arthroscopic surgery, no cast, crutches, or bed rest is required. Patients walk out of Cell Grand Clinic the same day. High-impact activities (running, jumping, heavy twisting) should be avoided for several weeks to let the cells settle in, but normal walking, work, and travel can resume immediately.
Is stem cell therapy for meniscus tears approved in Japan?
Yes. Japan’s Act on the Safety of Regenerative Medicine (2014) permits licensed clinics to deliver autologous stem cell therapy under MHLW oversight. Clinics must submit treatment plans to government-certified Special Committees for review and approval before treating any patient. Cell Grand Clinic holds 13 approved treatment plans and operates under ongoing government monitoring — a level of oversight unmatched in most countries offering stem cell therapy.
Is there an age limit?
No strict age limit exists. The 7-week culture protocol is especially important for older patients because it allows expansion of even slower-proliferating cells up to the full therapeutic dose of 100 million cells. Cell Grand Clinic has successfully treated patients well into their 70s and 80s for knee-related indications.
The Bottom Line
If you have a torn meniscus and you’re trying to decide what to do, here’s our honest summary:
No treatment is perfect. Surgery can relieve pain but comes with ~20% re-tear rates, recovery time, and long-term cartilage costs. Physical therapy alone often manages symptoms without changing much. And stem cell therapy — while not a magic eraser — is a real, regulated, joint-preserving option that helps a lot of patients skip or postpone surgery and get back to normal.
For the right candidate, it’s worth a serious look. And if you’re not the right candidate, we’ll tell you.
References
[1] Huang L, Zhang S, Wu J, Guo B, Gao T, Shah SZA, et al. Immunity-and-matrix-regulatory cells enhance cartilage regeneration for meniscus injuries: a phase I dose-escalation trial. Signal Transduct Target Ther. 2023;8(1):417. PMID: 37907503. https://doi.org/10.1038/s41392-023-01670-7
[2] Robinson J, Murray IR, Moatshe G, Chahla J, Tollefson LV, Parker DA, et al. Current practice of biologic augmentation techniques to enhance the healing of meniscal repairs: a collaborative survey within the Meniscus International Network (MenIN) Study Group. Knee Surg Sports Traumatol Arthrosc. 2025. https://doi.org/10.1002/ksa.12685
[3] Massey PA, Sampognaro G, Starnes E, Lowery MT, Duncan M, Sherman WF, et al. Improved outcomes after reinforced radial meniscus repair augmented with bone marrow aspirate concentrate. Arthrosc Sports Med Rehabil. 2023;5(3):e843-e851. https://doi.org/10.1016/j.asmr.2023.04.014
[4] Bąkowski P, Mieloch AA, Porzucek F, Mańkowska M, Ciemieniewska-Gorzela K, Naczk J, et al. Meniscus repair via collagen matrix wrapping and bone marrow injection: clinical and biomolecular study. Int Orthop. 2023;47(10):2409-2417. https://doi.org/10.1007/s00264-023-05711-2
[5] Cao M, Ou Z, Sheng R, Wang Q, Chen X, Zhang C, et al. Efficacy and safety of mesenchymal stem cells in knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Stem Cell Res Ther. 2025;16(1):122. https://doi.org/10.1186/s13287-025-04252-2
[6] Kim KI, Lee MC, Lee JH, Moon YW, Lee WS, Lee HJ, et al. Clinical efficacy and safety of the intra-articular injection of autologous adipose-derived mesenchymal stem cells for knee osteoarthritis: a phase III, randomized, double-blind, placebo-controlled trial. Am J Sports Med. 2023;51(9):2243-2253. https://doi.org/10.1177/03635465231179223
[7] Chen K, Aggarwal S, Baker H, Athiviraham A. Biologic augmentation of isolated meniscal repair. Curr Rev Musculoskelet Med. 2024;17(7):223-234. https://doi.org/10.1007/s12178-024-09898-8
[8] Kuczyński N, Boś J, Białoskórska K, Aleksandrowicz Z, Turoń B, Zabrzyńska M, et al. The meniscus: basic science and therapeutic approaches. J Clin Med. 2025;14(6):2020. https://doi.org/10.3390/jcm14062020
[9] Dai WL, Zhang H, Lin ZM, Shi ZJ, Wang J. Efficacy of platelet-rich plasma in arthroscopic repair for discoid lateral meniscus tears and meniscus failure rate after repair: a systematic review and meta-analysis. Orthop J Sports Med. 2022;9(10):23259671211038289. PMID: 34708138. https://doi.org/10.1177/23259671211038289
[10] Miller MD, Bansal S, Prabhakar A, Woolnough T, Haider MN, Ayeni OR, et al. All-inside versus inside-out meniscal repair in athletes: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2025. PMID: 40810129. https://doi.org/10.1002/ksa.12876
Evidence retrieved from PubMed. Meniscus repair failure rates vary substantially by tear type, patient age, surgical technique, and activity level; the 12–25% range cited in this article reflects the span from pooled meta-analytic data (~12% overall) up to higher-risk subgroups such as all-inside repair in athletes (~23%).
最終更新日:2026.04.17



 Inquiries by Email
Inquiries by Email

