Stem cell therapy for knee osteoarthritis uses mesenchymal stem cells to repair damaged cartilage and reduce inflammation — offering a non-surgical option when injections, hyaluronic acid, and physical therapy no longer help.
Below: how stem cells regrow cartilage, relief pain, what 5-year MRI follow-ups show, and how Japan’s MHLW-certified protocol compares to Mexican knee programs.
At Cell Grand Clinic in Osaka, Japan, cells cultured from your own minimal fat tissue over 7 weeks — up to 100 million autologous cells via intra-articular injection, never donor.
- Your Knee Pain Has a Third Option — Beyond Pills and Surgery
- What Causes Knee Osteoarthritis? Understanding Cartilage Loss
- Where Are You on the Scale? The Kellgren-Lawrence Grading System
How Does Stem Cell Therapy Repair Knee?
Is Stem Cell Therapy Effective for Knee Osteoarthritis? The 2025 Evidence- Why Adipose-Derived Stem Cells Outperform Other Sources
- How Many Stem Cells Do You Need for Knee OA? Why Dosage Matters
- Stem Cell Therapy vs. Knee Replacement: How Do They Compare?
- Why Cell Grand Clinic? What Is a “Grand Stem Cell”?
- Knee Stem Cell Treatment Process
- Who Is a Good Candidate for Knee Stem Cell Therapy?
- Postpone the knee replacement — repair the cartilage first.
- Frequently Asked Questions
- Conclusion: Restore Your Mobility — Naturally
- References
Your Knee Pain Has a Third Option — Beyond Pills and Surgery
Knee stem cell therapy is a non-surgical regenerative treatment that uses mesenchymal stem cells (MSCs) injected directly into the knee joint to reduce inflammation, support cartilage repair, and restore mobility — offering a scientifically validated alternative to knee replacement surgery.
Picture this. You’re in your 50s or 60s. You still want to play golf, hike mountain trails, or simply walk without pain. But every step reminds you that the cartilage in your knee is disappearing.
Your doctor back home has given you two choices: keep taking anti-inflammatory drugs and cortisone shots that mask the pain while the joint deteriorates — or schedule a total knee replacement when you can’t take it anymore.
Sound familiar? You’re not alone. Knee osteoarthritis (OA) affects roughly 650 million people aged 40 and older worldwide, and those numbers are climbing every year. It is one of the leading causes of disability on the planet.
But here’s what your orthopedic surgeon may not have told you: there is now a scientifically validated third option. A 2025 meta-analysis of 8 randomized controlled trials (502 patients) published in Stem Cell Research & Therapy confirmed that stem cell therapy for knee — without surgery — significantly reduces pain and improves function for at least 12 months.
Knee stem cell therapy is available today under Japan’s Ministry of Health, Labour and Welfare (MHLW) oversight, one of the world’s strictest regulatory frameworks for regenerative medicine. No surgery. No general anesthesia. No hospitalization. No long-term drug dependency.
What Causes Knee Osteoarthritis? Understanding Cartilage Loss
Knee osteoarthritis is a degenerative joint disease in which the protective cartilage covering the ends of the knee bones gradually breaks down, leading to bone-on-bone friction, chronic pain, swelling, and progressive loss of mobility.
Think of knee cartilage as a precision-engineered shock absorber. In a healthy joint, this smooth tissue lets the bones in your knee glide against each other with almost zero friction. It distributes your body weight evenly and protects the underlying bone from impact.
In osteoarthritis, this cartilage breaks down. Microscopic cracks appear on the surface, deepen over time, and eventually pieces break off entirely. When enough cartilage is lost, bone grinds directly against bone. That’s where the intense pain, swelling, stiffness, and grinding sensation (crepitus) come from.
The critical problem: adult cartilage cannot repair itself effectively. Unlike skin or bone, cartilage has no blood supply. Without blood flow, the body cannot deliver repair cells. Once cartilage damage begins, it tends to accelerate — creating a vicious cycle of inflammation, further loss, and escalating pain. This is precisely why regenerative approaches like stem cell therapy are being investigated as a way to break the cycle.
If you are experiencing chronic knee or joint pain, understanding this underlying mechanism is the first step toward exploring treatment options beyond conventional painkillers.
Where Are You on the Scale? The Kellgren-Lawrence Grading System
Doctors classify knee OA severity using the Kellgren-Lawrence (KL) system based on X-ray findings:
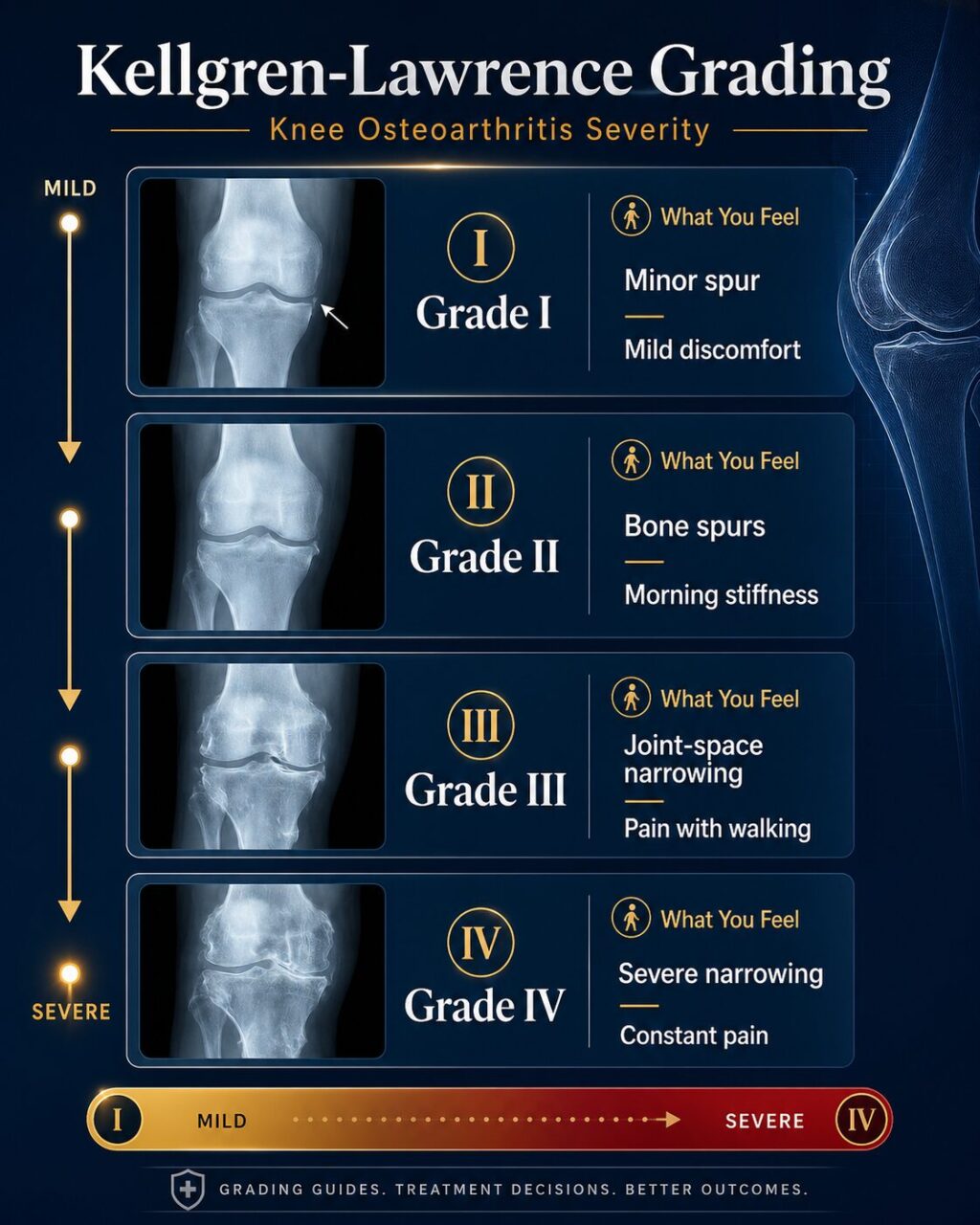
Stem cell therapy achieves the best outcomes for patients with KL Grade II–III OA, where sufficient cartilage remains for the stem cells to support repair. Even some Grade IV patients experience meaningful pain relief.
| Grade | X-Ray Findings | What You Feel | Stem Cell Benefit |
|---|---|---|---|
| 0 | Normal joint | No symptoms | — |
| I | Possible minor bone spur | Occasional mild discomfort after heavy activity | — |
| II ★ | Definite bone spurs, possible joint space narrowing | Pain during and after activity, morning stiffness | BEST |
| III ★ | Moderate narrowing, multiple spurs | Frequent pain, difficulty with stairs and prolonged walking | BEST |
| IV | Severe narrowing or absent joint space | Constant pain, severely limited mobility | Pain relief possible |
★ Grade II–III = The sweet spot for stem cell therapy. Enough cartilage remains for stem cells to support repair.
How Does Stem Cell Therapy Repair Knee?
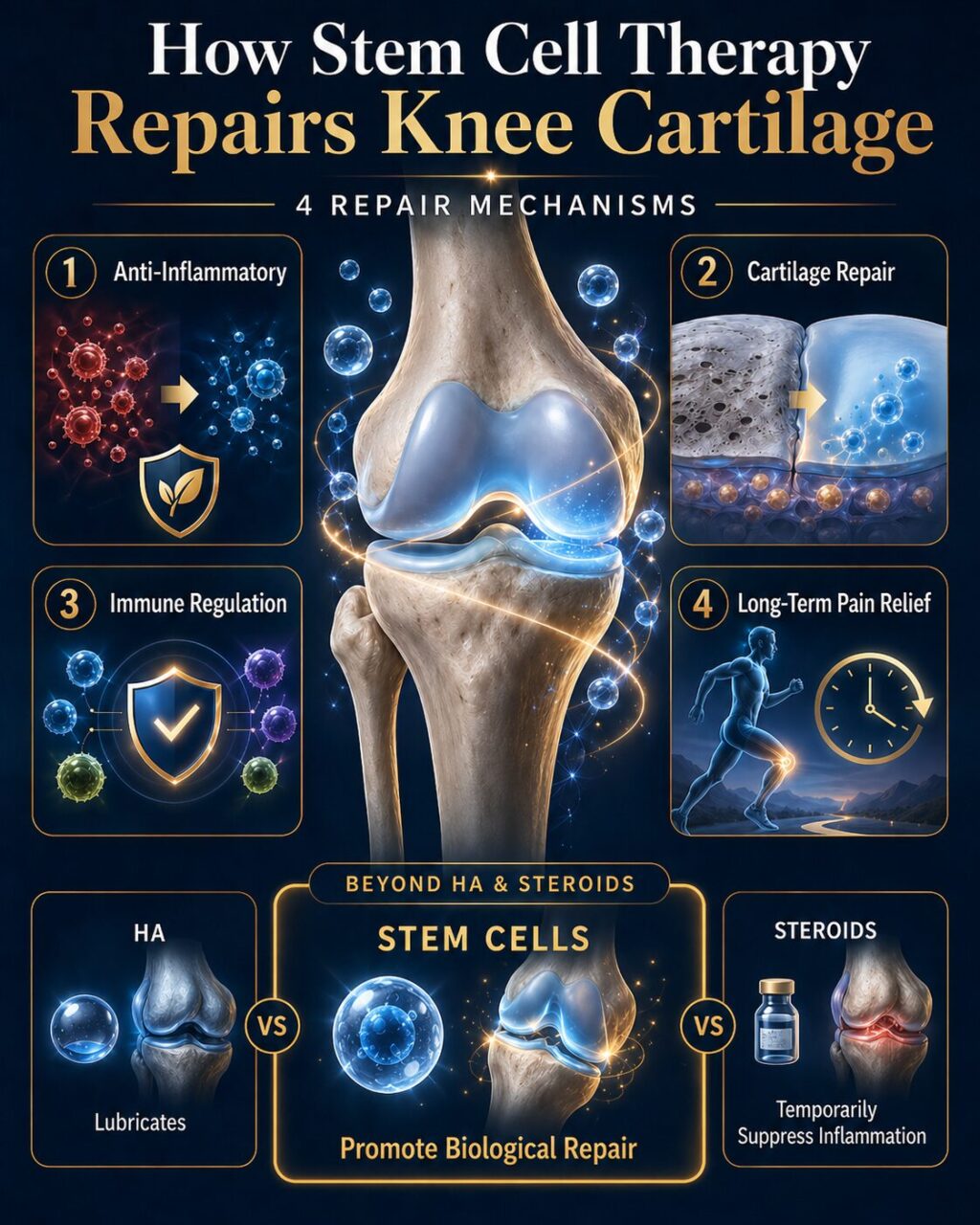
1. Anti-Inflammatory Action
Osteoarthritis involves chronic inflammation that actively destroys cartilage. MSCs secrete powerful anti-inflammatory molecules (IL-10, TGF-β, IL-1Ra) that work like internal fire extinguishers — they don’t just reduce today’s inflammation; they break the cycle causing ongoing damage.
2. Cartilage Protection and Repair
MSCs release growth factors that stimulate chondrocytes — the cells responsible for building and maintaining cartilage. Studies have documented measurable improvements in cartilage volume and quality following high-dose MSC injection.
3. Immune System Regulation
Through sophisticated paracrine signaling, MSCs regulate T cells, B cells, and macrophages within the joint [10]. This creates a microenvironment that favors tissue repair over destruction.
4. Sustained, Long-Term Pain Relief
By addressing inflammation, improving lubrication, and supporting tissue repair simultaneously, MSCs deliver pain relief that extends far beyond conventional injections. Clinical studies have documented sustained improvements at 12 months, 24 months, and up to 5 years post-treatment.
This is fundamentally different from a cortisone shot (wears off in weeks) or hyaluronic acid (lasts a few months at best). Stem cell therapy targets the root biological causes of your pain — not just the symptoms.
Is Stem Cell Therapy Effective for Knee Osteoarthritis? The 2025 Evidence
In March 2025, Cao et al. published a comprehensive systematic review in Stem Cell Research & Therapy, analyzing 8 randomized controlled trials (RCTs) involving 502 patients. This rigorous study focused exclusively on intra-articular Mesenchymal Stem Cells (MSC) injection without surgical intervention—providing the clearest evidence yet for stem cell therapy efficacy in knee osteoarthritis.
| Outcome Measure | 6-Month Results | 12-Month Results |
|---|---|---|
|
WOMAC Total Score (Overall knee function) |
Significant improvement (P = 0.01) |
Significant improvement (P = 0.03) |
|
Pain (VAS) (Pain intensity 0–100) |
−19.39 point reduction (P = 0.0008) |
−16.21 point reduction (P = 0.0003) |
|
KOOS Subscores (Knee function details) |
✓ Improved across all domains | ✓ Sustained improvement |
|
Adverse Events (Safety) |
No significant difference vs. control | Safe profile maintained |
In simple terms: researchers combined the results of 8 high-quality studies involving 502 real patients. They found that injecting stem cells into the knee — without any surgery — significantly reduced pain and made it easier to walk, bend, and use the knee normally. These benefits lasted at least one year, and the treatment was just as safe as doing nothing extra. Think of it like planting seeds in a damaged garden — the stem cells help your knee start repairing itself from the inside.
The meta-analysis concluded: “Intra-articular injection of MSCs alone could significantly improve knee pain and dysfunction in patients with unoperated OA.”
Further supporting these findings, Kim et al. published a Phase III, randomized, double-blind, placebo-controlled trial in The American Journal of Sports Medicine involving 261 patients — the largest individual RCT to date for knee stem cell therapy. Using 100 million adipose-derived stem cells, this study demonstrated significant efficacy in patients with Kellgren-Lawrence grade 3 knee OA.
Long-term data are also encouraging. A 5-year follow-up study by Kim et al. (2022), published in Stem Cells Translational Medicine, confirmed that improvements in pain and function were sustained up to 5 years after a single stem cell injection.
Reach us directly — WhatsApp and email inquiries are free of charge.
Why Adipose-Derived Stem Cells Outperform Other Sources
Not all stem cells are created equal. The 2025 meta-analysis conducted subgroup analyses comparing different cell sources—and the results were striking: Subgroup analysis revealed that adipose-derived MSCs produced statistically significant WOMAC improvement at both 6 months (P < 0.00001) and 12 months (P < 0.0001), while bone marrow-derived MSCs showed no significant improvement at either time point.
Adipose-Derived MSCs (ADMSCs) vs. Bone Marrow MSCs (BM-MSCs)
Why adipose tissue wins:
- Higher cell yield: Abdominal fat provides significantly more stem cells than bone marrow, especially critical in elderly patients
- Easier harvesting: Liposuction is minimally invasive compared to bone marrow aspiration from the iliac crest
- Superior proliferation: Adipose-derived cells demonstrate robust expansion during culture
- Lower donor-site morbidity: No risk of prolonged pain at the harvest site
At Cell Grand Clinic, we exclusively use autologous adipose-derived stem cells harvested from your own abdominal subcutaneous fat—leveraging the most effective cell source identified in clinical research.
| Outcome | Adipose (Fat) ✓ | Bone Marrow |
|---|---|---|
| 6-Month WOMAC |
Significant improvement P < 0.00001 |
No significant improvement P = 0.27 |
| 12-Month WOMAC |
Significant improvement P < 0.0001 |
No significant improvement P = 0.21 |
In simple terms: scientists compared two types of stem cells — ones taken from belly fat and ones taken from bone marrow (the spongy tissue inside your bones). The fat-derived stem cells worked significantly better at healing knees, with very strong statistical proof. The bone marrow cells did not show a clear benefit.
How Many Stem Cells Do You Need for Knee OA? Why Dosage Matters
High-dose stem cell therapy — defined as 100 million (1×10⁸) cells per injection — has been shown to produce significantly better outcomes for knee osteoarthritis than lower-dose protocols, according to the 2025 meta-analysis dose-response analysis.
The meta-analysis confirmed a clear dose-response relationship: high-dose MSC injection (1×10⁸ cells) produced significant improvement at both 6 and 12 months, while low-dose protocols showed no significant benefit at 6 months. The table below illustrates this critical difference.
| Time Point | High-Dose (100M) ✓ | Low-Dose (10–64M) |
|---|---|---|
| 6-Month WOMAC |
Significant improvement P = 0.002 |
No significant difference P = 0.16 |
| 12-Month WOMAC |
Significant improvement P < 0.0001 |
Marginal improvement P = 0.03 |
In simple terms: imagine trying to repair a large hole in a wall. If you use just a handful of plaster, it won’t do much. But if you use enough — 100 million stem cells — the repair is strong and lasts. Research proved that the high dose worked significantly better than smaller amounts, especially over 12 months.
Stem Cell Therapy vs. Knee Replacement: How Do They Compare?
Stem cell therapy and total knee replacement represent fundamentally different approaches to knee osteoarthritis: regenerative repair of the natural joint versus surgical replacement with an artificial prosthesis, each with distinct recovery timelines, risk profiles, and long-term outcomes.
Unlike knee replacement surgery, which requires months of rehabilitation and permanently removes the natural joint, stem cell therapy preserves the patient’s own knee structure with minimal downtime — most patients walk out of the clinic the same day. The following table compares the key differences between these treatment options.
| Feature | Stem Cell Therapy ★ | Hyaluronic Acid | Knee Replacement |
|---|---|---|---|
| Primary Goal | Regeneration & Repair | Temporary Lubrication | Artificial Replacement |
| Invasiveness | Injection only | Injection | Major surgery |
| Recovery Time | Walk same day | None | 3–6 months rehab |
| Duration of Effect | Years | Weeks to months | 15–20 years (prosthesis) |
| Your Natural Joint | ✓ Preserved | ✓ Preserved | ✗ Removed |
| Serious Risks | Minimal (your own cells) | Low | Infection, blood clots, implant failure |
As the comparison shows, stem cell therapy offers a unique balance of regenerative potential, minimal invasiveness, and preservation of the natural joint — making it an attractive option for patients who want to avoid or delay surgery.
Why Cell Grand Clinic? What Is a “Grand Stem Cell”?
Cell Grand Clinic is an MHLW-certified regenerative medicine clinic in Osaka, Japan, that delivers high-dose autologous adipose-derived stem cell therapy under one of the world’s most rigorous government oversight frameworks — with over 3,000 treatments performed.
Quality That Delivers Results
Many clinics offer “stem cell therapy,” but the term alone tells you nothing about quality. A treatment using poorly cultured, low-count, or aged cells is fundamentally different from one using fresh, verified, high-dose stem cells — even though both carry the same name.
That’s why Cell Grand Clinic created a proprietary quality standard: the “Grand Stem Cell.” Only cells that pass all four of the following criteria earn this designation — and a Certificate of Quality is issued to every patient as proof.
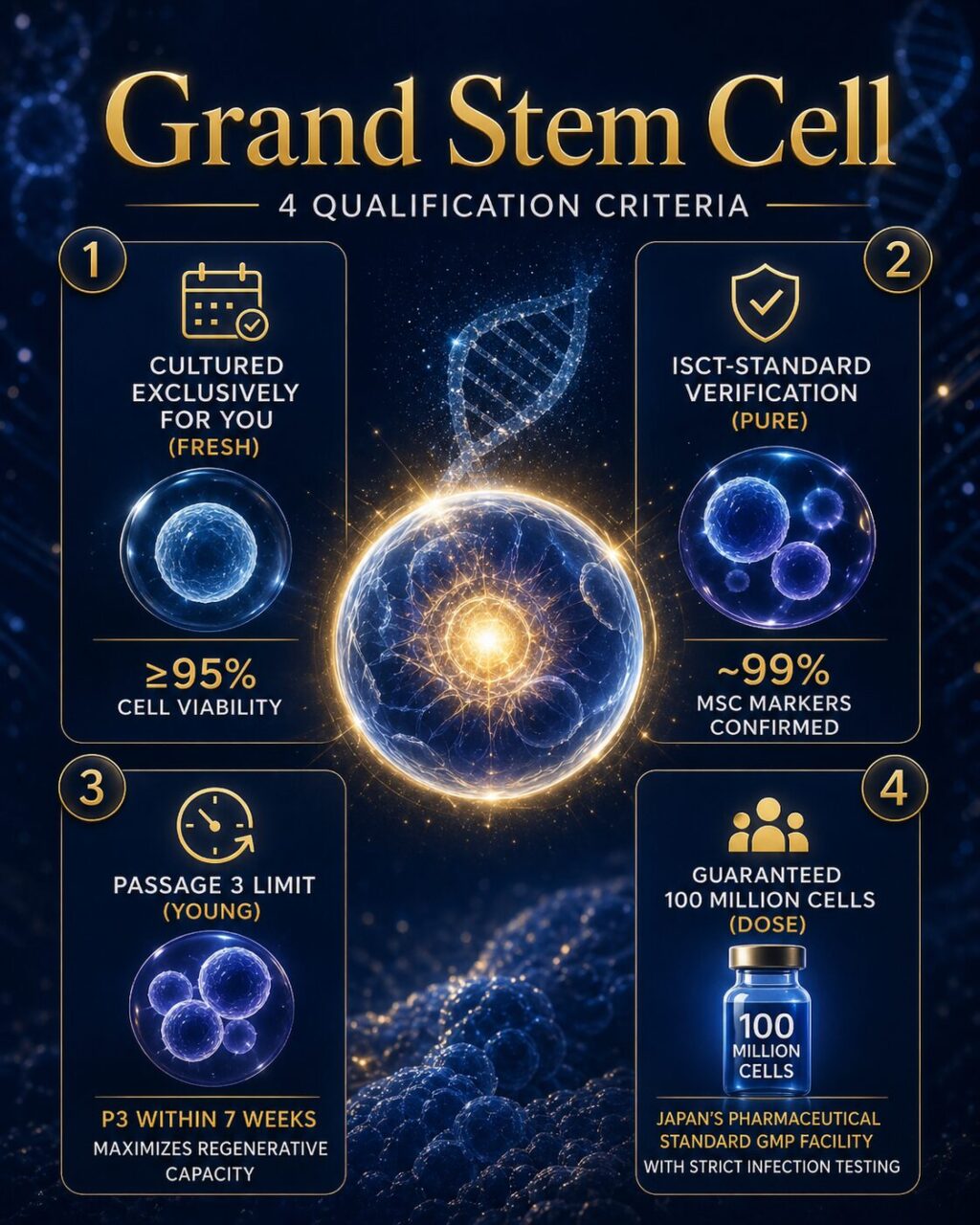
1. Cultured Exclusively for You (Fresh)
Grand Stem Cells are never stockpiled or pre-made. Culture begins only after your treatment is confirmed, with cells expanded over a dedicated 7-week period. Cell viability of ≥95% is verified before every administration — compared to an industry average of 70–85%. Dead or dying cells cannot heal.
2. ISCT-Standard Verification (Pure)
Every batch undergoes surface antigen testing per International Society for Cell & Gene Therapy (ISCT) global standards. Approximately 99% CD73+, CD90+, CD105+ expression is confirmed; contaminants (CD45−, CD34−) are eliminated. This proves each administered cell is a genuine mesenchymal stem cell.
3. Passage 3 Limit (Young)
Stem cells age with each round of culture expansion. Cell Grand Clinic enforces a strict Passage 3 (P3) limit within the 7-week culture, keeping cells biologically young and maximizing their regenerative capacity. Over-expanded cells lose their repair ability and may compromise safety.
4. Guaranteed 100 Million Cells (Dose)
Every patient receives a guaranteed dose of 100 million cells per administration, processed at a GMP-compliant Cell Processing Center (CPC) certified under Japanese pharmaceutical standards. The 2025 meta-analysis showed that high-dose (≥100M) significantly outperforms lower doses — insufficient cells mean insufficient results.
Cell Quality: The Key Factor That Impacts Stem Cell Therapy Outcomes Learn MoreIn Simple Terms: Think of it like baking a cake. You need fresh ingredients (not expired ones), the right ingredients (not substitutes), ingredients that are at their best (not stale), and enough of them (not half a recipe). “Grand Stem Cell” means every single one of these quality checks is passed — and you get a certificate to prove it.
All cells are processed at a GMP-compliant Cell Processing Center (CPC) certified under Japanese pharmaceutical standards — the same level of quality control applied to manufactured drugs.
Japanese Government Approval: The World’s Gold Standard
Japan’s Act on the Safety of Regenerative Medicine (2014, regularly updated) created one of the world’s first comprehensive legal frameworks for stem cell treatments. Clinics must submit detailed treatment plans to MHLW-certified committees for review and approval.
13 certified treatment plans (Type II and III licenses) —
Cell Grand Clinic holds One of the broadest portfolios of any regenerative medicine clinic in Japan. Our knee OA treatment is not “experimental” or “off-label.” It is a government-regulated medical treatment.
Compare this to the situation in many other countries, where stem cell clinics operate with little regulatory oversight, using uncharacterized cell preparations with no quality control requirements.

Academic Partnership and Expert Oversight
Our cell culture protocols are developed in collaboration with Professor Takahiro Ochiya’s research team, whose work on exosomes and stem cell biology has appeared in top-tier journals. Every treatment plan is personally overseen by Dr. Yuichi Wakabayashi, Diplomate of the American Board of Regenerative Medicine, ensuring care that meets the highest international standards.
Reach us directly — WhatsApp and email inquiries are free of charge.
Knee Stem Cell Treatment Process
We’ve optimized our protocol for busy professionals and international visitors:
Share your MRI/X-ray results with our medical team for preliminary assessment.
Minimally invasive fat harvest (~30 minutes) under local anesthesia. Return to your hotel immediately.
⚡ Don’t wait 7 weeks in pain
During Visit 1, your doctor may also administer Exosome Therapy or PRP (Platelet-Rich Plasma) injection directly into your knee. These treatments provide immediate anti-inflammatory and pain-relieving effects — helping you manage your current knee pain while your stem cells are being cultured over the next 7 weeks. Think of it as a “bridge treatment” that starts working right away, so you don’t have to endure unnecessary discomfort while waiting for your personalized stem cells to be ready.
Your cells are processed and expanded at our specialized CPC. Return home while we cultivate your cells to therapeutic levels — 100 million Grand Stem Cells.
✓ If you received Exosome/PRP in Visit 1, these continue working to relieve pain during this waiting period.
Return to Osaka for stem cell administration via direct intra-articular injection (~30 minutes including preparation).
Note: Repeat treatments may be recommended depending on disease severity.
Alternatives to Knee Replacement: PRP, HA, MSC & Exosome Compared Compare Options Exosome Therapy: Same-Day, Cell-Free Regeneration Learn MoreWho Is a Good Candidate for Knee Stem Cell Therapy?
This therapy is particularly effective for:
- Kellgren-Lawrence Grade II-III OA: Patients with moderate degeneration typically see the best outcomes
- Active individuals: Those who want to continue sports (golf, tennis, hiking) that may become impossible after joint replacement
- Surgery-averse patients: Those who cannot or prefer not to undergo general anesthesia and prolonged rehabilitation
- Patients seeking to delay surgery: Even in advanced cases, stem cell therapy can reduce pain and potentially postpone surgical intervention
Postpone the knee replacement — repair the cartilage first.
Stem cell therapy — from $19,800 USD per session
$19,800 USD includes:
- 100 million autologous ADSCs
- 7-week culture at MHLW-certified CPC · passage ≤3
- Quality certificate · 95%+ viability
- 1 / 3 / 6 month remote follow-up
Final pricing depends on your individual case:
- Combination protocols
- Multiple sessions
Have Dr. Wakabayashi review your case.
Share your medical history and current treatments. Dr. Wakabayashi reviews each international inquiry and responds with a written feasibility note, recommended protocol, and quote. Typical turnaround: 1–3 business days.
Frequently Asked Questions
How does stem cell therapy compare to total knee replacement?
Knee replacement surgery removes your natural joint and replaces it with artificial components, requiring 3–6 months of rehabilitation. Stem cell therapy preserves and repairs your natural joint through injection, with patients walking out the same day. For appropriate candidates, stem cell therapy can significantly reduce pain and improve mobility, potentially eliminating or delaying the need for surgery. Cell Grand Clinic offers a remote consultation to help determine which approach is appropriate for your specific condition.
Is there an age limit for knee stem cell therapy?
No strict age limit exists for stem cell therapy. Cell Grand Clinic has successfully treated patients in their 80s. The 7-week culture protocol is especially important for older patients, as it allows expansion of even cells with reduced proliferative capacity to the full therapeutic dose of 100 million cells.
Can I walk immediately after treatment?
Yes. Unlike surgery, no cast or crutches are required. Patients walk out of Cell Grand Clinic after the injection. Avoiding high-impact activities for several weeks is recommended to allow cellular integration, but normal daily activities can be resumed immediately.
How long does stem cell therapy for knee OA last?
Clinical studies have shown sustained improvements at 12 months and beyond. A 5-year follow-up study published in Stem Cells Translational Medicine confirmed that pain relief and functional improvement were maintained for up to 5 years after a single injection. Individual results vary based on OA severity, activity level, and overall health.
Is knee stem cell therapy safe?
The 2025 meta-analysis of 502 patients found no significant difference in adverse events between stem cell and control groups. Since Cell Grand Clinic uses autologous cells (your own tissue), immune rejection is not a concern. The most common side effects are temporary pain and swelling at the injection site, which typically resolve within days.
How many stem cells are injected for knee osteoarthritis?
The 2025 meta-analysis showed that high-dose regimens — defined as 100 million cells or more — produced significantly greater pain relief than lower doses. Cell Grand Clinic guarantees a dose of 100 million fresh, quality-verified Grand Stem Cells cultured over a 7-week period, ensuring every patient receives the clinically optimal amount.
Is stem cell therapy for knee OA approved in Japan?
Yes. Japan has a dedicated legal framework — the Act on the Safety of Regenerative Medicine (2014) — that permits licensed clinics to offer stem cell therapy under government oversight. Clinics must submit a treatment plan to an MHLW-certified Special Committee, pass safety and ethics reviews, and receive an official plan number before treating patients. Cell Grand Clinic holds 13 approved treatment plans and operates under continuous government monitoring.
What is the difference between stem cell therapy and PRP for knee OA?
PRP (Platelet-Rich Plasma) concentrates growth factors from your blood to reduce inflammation, but it does not contain stem cells and cannot regenerate cartilage tissue. Stem cell therapy delivers living mesenchymal stem cells that can differentiate into cartilage-forming cells, secrete anti-inflammatory and regenerative factors, and potentially repair damaged tissue at a structural level. Clinical evidence suggests stem cell therapy produces greater and longer-lasting improvements, particularly for moderate-to-severe osteoarthritis (KL Grade II–III).
How much does knee stem cell therapy cost in Japan?
The cost of stem cell therapy in Japan varies depending on the cell count, culture duration, and quality control standards used. Premium treatments with high cell counts (100 million+) and rigorous quality certification typically range from approximately $15,000–$50,000 USD per session. Cell Grand Clinic provides a personalized cost estimate during the initial consultation — contact us via WhatsApp or email for details.
How long does it take to see results from knee stem cell therapy?
Most patients begin to notice improvement within 1–3 months after stem cell injection, as the cells reduce inflammation and begin supporting cartilage repair. The 2025 meta-analysis documented statistically significant improvement at the 6-month mark, with benefits continuing to strengthen through 12 months. Full therapeutic effects develop gradually as the stem cells modulate the joint environment over time.
Conclusion: Restore Your Mobility — Naturally
For patients with moderate knee osteoarthritis seeking a non-surgical alternative, high-dose adipose-derived stem cell therapy offers a scientifically validated option. The 2025 meta-analysis confirmed that 100 million autologous MSCs injected into the knee significantly reduce pain and improve function for at least 12 months, with a safety profile comparable to placebo.
Cell Grand Clinic offers this scientifically validated, non-surgical solution for knee osteoarthritis — delivered under Japan’s MHLW oversight with guaranteed quality through the Grand Stem Cell standard.
Reach us directly — WhatsApp and email inquiries are free of charge.
References
Cui AY, Li HZ, Wang DW, Zhong JL, Chen YF, Lu HD. Global, regional prevalence, incidence and risk factors of knee osteoarthritis in population-based studies. EClinicalMedicine. 2020;29-30:100587. https://doi.org/10.1016/j.eclinm.2020.100587
Cao M, Ou Z, Sheng R, Wang Q, Chen X, Zhang C, et al. Efficacy and safety of mesenchymal stem cells in knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Stem Cell Res Ther. 2025;16(1):122. https://doi.org/10.1186/s13287-025-04252-2
Kim KI, Lee MC, Lee JH, Moon YW, Lee WS, Lee HJ, et al. Clinical efficacy and safety of the intra-articular injection of autologous adipose-derived mesenchymal stem cells for knee osteoarthritis: a phase III, randomized, double-blind, placebo-controlled trial. Am J Sports Med. 2023;51(9):2243-2253. https://doi.org/10.1177/03635465231179223
Kim KI, Lee WS, Kim JH, Bae JK, Jin W. Safety and efficacy of the intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritic knee: a 5-year follow-up study. Stem Cells Transl Med. 2022;11(6):586-596. https://doi.org/10.1093/stcltm/szac024
Song N, Scholtemeijer M, Shah K. Mesenchymal stem cell immunomodulation: mechanisms and therapeutic potential. Trends Pharmacol Sci. 2020;41(9):653-664. https://doi.org/10.1016/j.tips.2020.06.009
Copp G, Robb KP, Viswanathan S. Culture-expanded mesenchymal stromal cell therapy: does it work in knee osteoarthritis? A pathway to clinical success. Cell Mol Immunol. 2023;20(6):626-650. https://doi.org/10.1038/s41423-023-01020-1
Huang ZY, Zhang S, Cao MD, Lin ZJ, Kong L, Wu X, et al. What is the optimal dose of adipose-derived mesenchymal stem cells treatment for knee osteoarthritis? A conventional and network meta-analysis of randomized controlled trials. Stem Cell Res Ther. 2023;14(1):245. https://doi.org/10.1186/s13287-023-03475-5
Maheshwer B, Polce EM, Paul K, Williams BT, Wolfson TS, Yanke A, et al. Regenerative potential of mesenchymal stem cells for the treatment of knee osteoarthritis and chondral defects: a systematic review and meta-analysis. Arthroscopy. 2021;37(1):362-378. https://doi.org/10.1016/j.arthro.2020.05.037
Thompson M, Mei SHJ, Wolfe D, Champagne J, Fergusson D, Stewart DJ, et al. Cell therapy with intravascular administration of mesenchymal stromal cells continues to appear safe: an updated systematic review and meta-analysis. EClinicalMedicine. 2020;19:100249. https://doi.org/10.1016/j.eclinm.2019.100249
Updated: 2026.05.16



 Inquiries by Email
Inquiries by Email

