If your knee hurts every time you walk up stairs, every morning when you get out of bed, or every time the weather changes — you already know that “just take it easy” is not an answer. You have probably tried braces, ice, physical therapy, maybe hyaluronic acid injections or a steroid shot. And at some point, the conversation with your orthopedic surgeon drifts toward total knee replacement.
Before you agree to surgery, it is worth knowing what else exists in 2026 — and being honest about what each option can and cannot do. Knee replacement is a genuinely life-changing operation for severe arthritis, but it involves hospital stays, months of rehabilitation, and a small but real risk of infection and reoperation. At the same time, conservative options are not all equal: hyaluronic acid, platelet-rich plasma (PRP), and stem cell therapy rest on very different evidence bases.
This article compares all four — total knee replacement, hyaluronic acid, PRP, and stem cell therapy — using only peer-reviewed human data published through 2025. The goal is not to push one answer. It is to help you talk to your doctor from a position of real information.
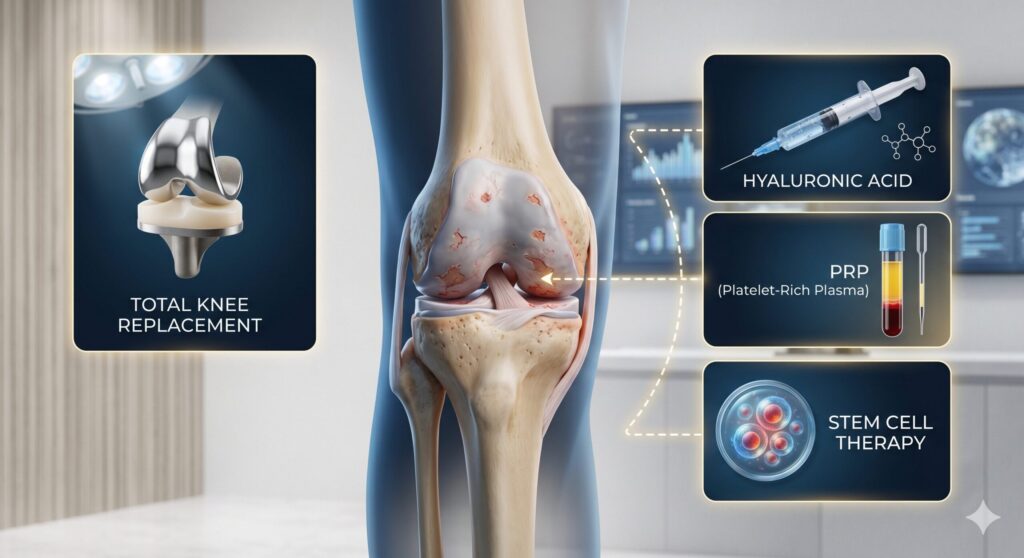
- What Knee Osteoarthritis Really Is — And Why It Gets Worse
- Your Full Range of Options, at a Glance
- Total Knee Replacement (TKA): The Gold Standard, With Real Trade-offs
- Hyaluronic Acid (HA): Short-Term Lubrication, Limited Reach
- Platelet-Rich Plasma (PRP): Better Than HA, But the Evidence Is Mixed
- Stem Cell Therapy for the Knee: The Leading Non-Surgical Option in 2026
- Lifestyle: The Foundation Under Every Option
- Side-by-Side Comparison
- Am I a Candidate for Stem Cell Therapy?
- Why Japan, and Why Cell Grand Clinic
- Frequently Asked Questions
- Closing
- References
- Image Suggestions (for insertion during WordPress upload)
What Knee Osteoarthritis Really Is — And Why It Gets Worse
Knee osteoarthritis (OA) is a slow, progressive disease in which the cartilage that cushions the joint breaks down, the underlying bone thickens, and inflammation spreads through the joint lining. The result is pain, stiffness, swelling, and eventually difficulty walking. It is the single most common cause of chronic knee pain in adults over 50.
A 2021 JAMA review published in JAMA described knee OA as one of the leading global causes of disability, with symptoms typically worsening over years rather than months — meaning most patients have a meaningful window of time to consider both surgical and non-surgical options before the joint reaches an end stage. The same review emphasized that no drug has yet been shown to stop or reverse the underlying cartilage loss; current medications mainly manage pain.
Meniscus tears, often caused by a twisting injury or by gradual wear, make things worse by removing the shock-absorbing cushion between the femur and tibia. Both conditions — OA and meniscus damage — share the same fundamental problem: the joint’s repair capacity is limited, and without intervention, the tissue damage tends to accumulate.
Your Full Range of Options, at a Glance
Before going deep on each treatment, it helps to see them side by side. The main options for knee OA and meniscus injury fall into three tiers: lifestyle and physical therapy, injection-based regenerative treatments (hyaluronic acid, PRP, stem cells), and surgery (arthroscopy, osteotomy, partial or total knee replacement). Each has a different goal, a different invasiveness, and a very different evidence base.
The honest framing: when the joint has collapsed and the bone is rubbing on bone, surgery is often the right answer. When the joint still has cartilage to work with, biologic treatments — especially stem cell therapy — can meaningfully change the disease trajectory. Lifestyle measures underpin every other treatment at every stage.
Total Knee Replacement (TKA): The Gold Standard, With Real Trade-offs
What TKA is, in 60 words
Total knee arthroplasty (TKA) is major orthopedic surgery in which the damaged cartilage and bone surfaces of the knee are removed and replaced with metal and plastic components. It is designed to eliminate pain and restore function in end-stage arthritis. For severe osteoarthritis with bone-on-bone changes, TKA remains the most effective intervention available — it is the only treatment that fully removes the diseased tissue.
The honest numbers
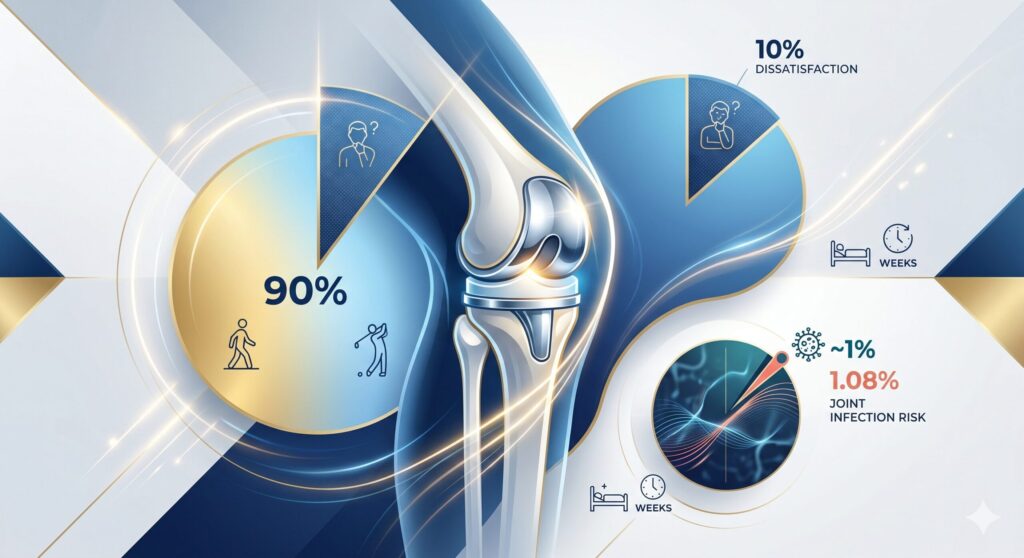
A 2022 systematic review in the Journal of Arthroplasty synthesizing data on patient-reported outcomes after TKA found that the average rate of dissatisfaction was approximately 10%, and 7.3% when complications were excluded — meaning roughly one in ten people who undergo TKA do not get the outcome they hoped for, even when the operation is technically successful. The most common drivers were age under 65, depression or anxiety, pain catastrophizing, and unmet expectations.
A 2024 meta-analysis published in the Journal of Orthopaedic Surgery and Research estimated that periprosthetic joint infection (PJI) occurs in roughly 1.08% of primary TKAs — a serious complication that often requires a second surgery to wash out the joint, remove the implant, and reimplant a new one over a months-long process. This is uncommon, but when it happens, it is one of the most difficult complications in orthopedic surgery.
TKA also involves a typical 3 to 5-day hospital stay, 4 to 12 weeks of intensive rehabilitation before returning to normal daily activity, and implant longevity of roughly 15–20 years — meaning younger patients may face a revision surgery later in life, which is technically more difficult than the first operation.
None of this means knee replacement is the wrong choice. For severe, bone-on-bone arthritis with deformity and function loss, TKA delivers results that no non-surgical therapy can match. The question is simply whether you are there yet.
Hyaluronic Acid (HA): Short-Term Lubrication, Limited Reach
What HA does, in 60 words
Hyaluronic acid injections (“gel shots”) add a viscous fluid similar to the knee’s natural synovial fluid, improving joint lubrication and cushioning for several weeks to months. HA does not repair cartilage or change the underlying disease. It is useful for short-term symptom relief in mild to moderate osteoarthritis, but effects typically fade, and guidelines now consider HA a modest — not essential — option.
A 2021 randomized trial of 200 patients published in BMC Musculoskeletal Disorders compared HA, PRP, PRGF, and ozone injections in knee OA. All four groups improved in the short term, but HA showed the smallest and least durable effect compared with the platelet-based options. In everyday language: HA tends to work, but not for long, and the newer biologic treatments tended to outperform it.
HA’s main strength is its safety profile and minimal downtime — the risks are small and the procedure is quick. Its main limitation is that it is purely symptomatic. After the fluid is absorbed, nothing structural has changed.
Platelet-Rich Plasma (PRP): Better Than HA, But the Evidence Is Mixed
What PRP is, in 60 words
Platelet-rich plasma is prepared by drawing a small amount of the patient’s blood, spinning it in a centrifuge to concentrate the platelets, and injecting the concentrate back into the joint. Platelets release growth factors that reduce inflammation and, in some studies, support cartilage metabolism. PRP is an autologous biologic — your own blood — and sits a step above HA in most head-to-head comparisons.
Why the evidence is genuinely mixed
A landmark 2021 JAMA trial called RESTORE, published in JAMA, randomized 288 patients with mild to moderate knee OA to three monthly PRP injections or saline placebo. At 12 months, there was no significant difference in pain reduction or cartilage volume between the two groups. Put simply: in this well-conducted trial, PRP worked about the same as salt water.
A 2023 network meta-analysis of 35 studies covering 3,104 patients, published in BMC Musculoskeletal Disorders, came to a different conclusion. It ranked PRP and PRP combined with hyaluronic acid (PRP+HA) above hyaluronic acid alone and corticosteroid for both pain (VAS) and function (WOMAC) at 6 and 12 months — and reported no increase in treatment-related adverse events. In practical terms, when pooled across many studies with varying protocols, PRP tended to outperform HA and steroid for most patients over 6–12 months.
How can both be true? PRP is not a single product. Preparation method, platelet concentration, volume, number of injections, and whether leukocytes are included all vary widely between studies — and the results vary accordingly. The honest reading is that PRP often helps, but results are less predictable than patients are sometimes told, and in rigorously blinded trials its benefit over placebo can shrink.
Stem Cell Therapy for the Knee: The Leading Non-Surgical Option in 2026
What stem cell therapy is, in 60 words
Stem cell therapy for the knee uses mesenchymal stem cells — most often adipose-derived stem cells (ADSCs) harvested from the patient’s own fat — to modulate joint inflammation, signal repair in cartilage cells, and support the joint environment. Unlike HA or PRP, stem cells release a continuous cocktail of growth factors and anti-inflammatory molecules for weeks, making them the most biologically active of the non-surgical options.
Two different reasons to consider stem cells
It helps to split the conversation in two, because the goal of treatment depends on where your knee is today.
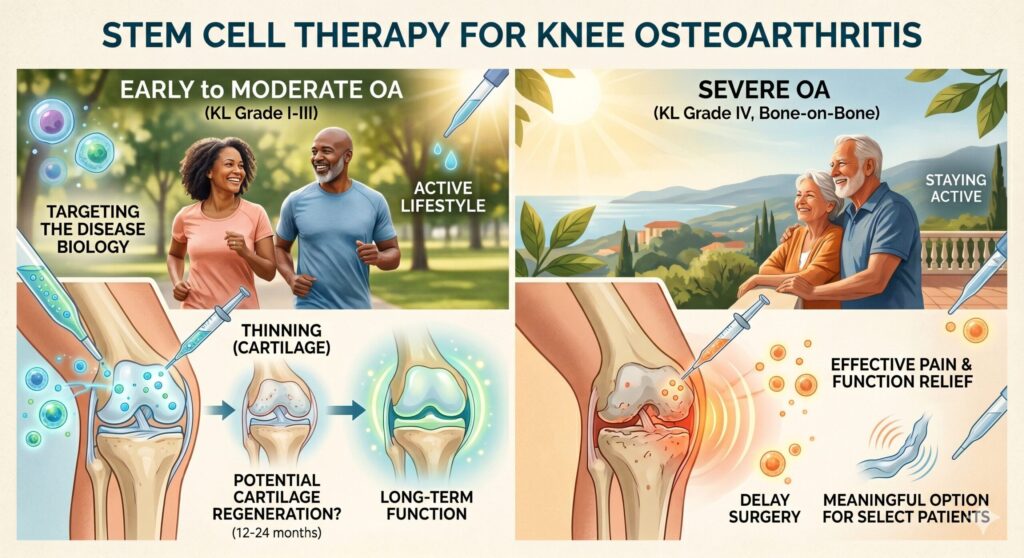
For mild to moderate osteoarthritis (roughly KL Grade I–III) — where cartilage is thinning but the joint has not yet collapsed — stem cell therapy is the one non-surgical option that aims at the underlying biology of the disease, not just its symptoms. Published trials in this population show sustained improvements in pain, function, and, in some imaging studies, cartilage signal at 12–24 months. For a well-selected early-stage patient, the realistic hope is not only pain relief but meaningful, durable recovery of joint function — a trajectory closer to disease modification than to symptom management.
For severe, end-stage osteoarthritis (KL Grade IV, bone-on-bone) — where the cartilage is effectively gone — stem cell therapy will not rebuild a lost joint surface. What it still offers, however, is the most effective pain and function relief available among non-surgical options. For patients who cannot undergo surgery because of age, comorbidities, anticoagulation, or personal preference — or who want to delay a replacement for a year or two — ADSC therapy remains a meaningful tool, even when the ceiling of improvement is lower than in earlier-stage disease.
For patients with a specific existing knee OA column at Cell Grand Clinic, a more condition-focused overview is available in our dedicated page on stem cell therapy for knee osteoarthritis and knee pain.
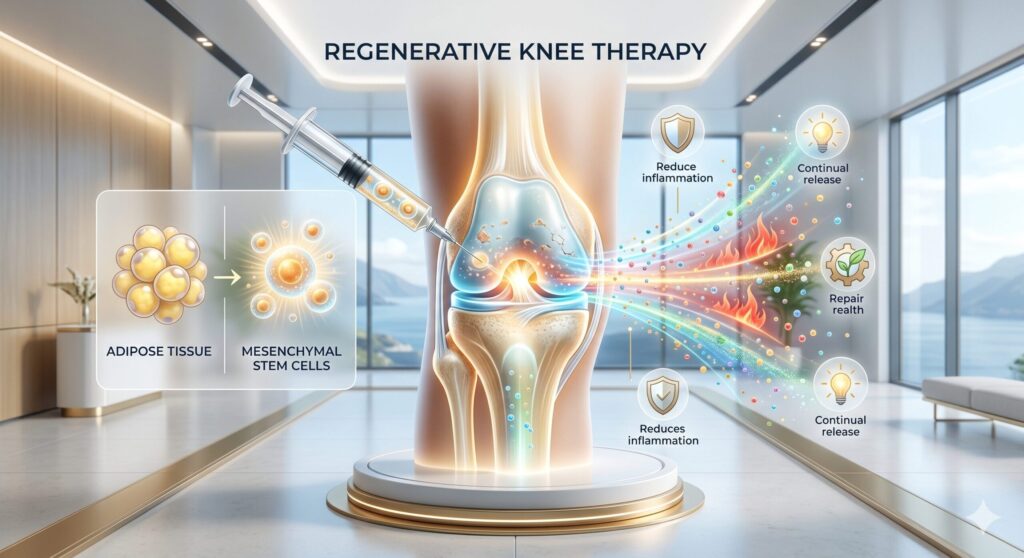
How stem cells actually work in a knee joint
A 2021 review in Frontiers in Immunology summarized the use of mesenchymal stem cells in arthritic disease and characterized their mechanism as primarily anti-inflammatory, immunomodulatory, and regenerative — meaning the cells work by signaling, not by becoming new cartilage themselves. They release growth factors and cytokines that shift the joint from a pro-inflammatory state to a pro-repair state, allowing the patient’s own tissue to recover. For a reader, the mental picture is closer to “reset the thermostat of the joint” than “paste in new cartilage.”
The strongest clinical evidence
Of all the non-surgical options, stem cell therapy has the most encouraging recent meta-analytic data. A 2025 systematic review and meta-analysis of 8 randomized controlled trials (n=502) published in Stem Cell Research & Therapy reported that intra-articular MSC injection produced a 10.31-point greater WOMAC improvement than placebo at 12 months (95% CI 0.96–19.67; P=0.03), with VAS and KOOS scores also improving at 6 and 12 months and the adipose-derived subgroup showing the strongest effect. The subgroup comparison matters because Cell Grand Clinic uses adipose-derived stem cells, which is the exact cell type that performed best in this pooled analysis.
Taken together, these trials describe a consistent pattern: adipose-derived stem cell therapy improves pain and function in knee OA more than placebo, more than hyaluronic acid in head-to-head comparisons, and with a safety profile that has been acceptable across more than a decade of published data. This is why, among non-surgical options, stem cell therapy has become the leading choice for patients who want to address the joint biology and not just the symptoms.
What about meniscus injuries?
Meniscus tears historically had only two answers: partial meniscectomy (remove the torn tissue) or, rarely, meniscus repair. A 2023 systematic review in Tissue Engineering Part B: Reviews evaluated 72 preclinical and 6 clinical studies of cell-based meniscus regeneration and concluded that mesenchymal stem cells (especially bone marrow-derived MSCs) are the most commonly studied cell type, with combinatorial strategies — pairing cells with scaffolds, biomaterials, or co-culture — showing greater promise for tissue repair than single approaches. A 2023 Phase I dose-escalation trial in Signal Transduction and Targeted Therapy using immunity-and-matrix-regulatory cells (IMRCs) derived from human embryonic stem cells, in 18 patients with meniscus injury, found intra-articular IMRC injection to be safe over 12 months, with MRI evidence of meniscus repair and improved knee functional scores — early but encouraging evidence that regenerative approaches can extend beyond OA into meniscus pathology.
For a closer look at how regenerative medicine is being applied specifically to torn meniscus, see our dedicated guide on stem cell therapy for torn meniscus repair.
Lifestyle: The Foundation Under Every Option
No injection, biologic, or surgical implant performs as well as it could without the basics. A 2022 randomized trial of 823 adults with knee OA and overweight or obesity, published in JAMA, showed that a combined diet-plus-exercise program produced a small but statistically significant pain reduction over 18 months compared with an attention-control group — an adjusted between-group difference of 0.6 points on the WOMAC pain scale (range 0–20), with the intervention group losing on average 7.7 kg of body weight versus 1.7 kg in controls. The pain difference is modest, but the intervention has no side effects, no cost beyond effort, and compounds with every other treatment.
A practical rule: every 1 kg of weight lost reduces knee joint loading with each step by about 4 kg. Combining weight management with quadriceps strengthening is still the single best thing most patients can do between any injection or procedure.
Side-by-Side Comparison
The table below summarizes how the main options compare across the dimensions patients most often ask about — recovery time, regenerative potential, evidence strength, and typical cost range. It is meant as an orientation, not a prescription: the right choice depends on KL grade, symptoms, age, activity goals, and comorbidities.
| Option | Mechanism | Invasiveness | Evidence | Best for |
|---|---|---|---|---|
| Lifestyle / PT | Load reduction, muscle support | None | Strong (JAMA RCT) | All stages, as a foundation |
| Hyaluronic acid | Joint lubrication | Minimal (injection) | Modest, short-duration | Mild OA, short-term relief |
| PRP | Growth-factor signaling | Minimal (injection) | Mixed (negative RCT, positive network MA) | Mild–moderate OA, trial of biologic |
| Stem cells (ADSC) | Paracrine signaling, immunomodulation | Minor (fat harvest + injection) | Strongest among non-surgical (2025 MA) | Mild–severe OA wishing to avoid/delay surgery |
| Knee replacement | Prosthetic replacement of joint | Major surgery | Strong; ~10% dissatisfaction | End-stage, bone-on-bone OA |
This is a general comparison. Individual suitability depends on disease stage, imaging, comorbidities, and patient goals. Consultation is required before any decision.
Am I a Candidate for Stem Cell Therapy?
The patients who tend to benefit most from ADSC therapy for the knee share a recognizable profile. The clinical framing at Cell Grand Clinic looks like this:
Individual suitability is determined during a medical consultation. This checklist is for general reference only.
Patients with end-stage, bone-on-bone deformity and severe mechanical malalignment may still need surgery. Stem cell therapy is not a replacement for TKA when the joint has structurally collapsed — it is a different tool, used earlier or in combination.
Why Japan, and Why Cell Grand Clinic
Japan is one of the few countries in the world where regenerative medicine is regulated directly by the national government. Under the Act on the Safety of Regenerative Medicine, every clinic providing cell-based therapy must be certified by the Ministry of Health, Labour and Welfare (MHLW), the equivalent of the FDA. This is not a voluntary accreditation; it is a legal requirement with government oversight of cell handling, facility standards, and informed consent.
Cell Grand Clinic operates within this framework. Treatments use the patient’s own adipose-derived stem cells (autologous ADSCs), processed in a CPC-grade (Cell Processing Center) laboratory, under a physician with NIH training and experience across more than 3,000 patient cases.
| Factor | Japan (MHLW-certified) | Mexico | Thailand |
|---|---|---|---|
| Government Oversight | MHLW certified ✓ | Limited regulation | Emerging regulation |
| Cell Quality Standards | CPC-grade, 7-week culture ✓ | Varies by clinic | Varies by clinic |
| Cell Count per Treatment | Up to 200 million ✓ | Varies | Varies |
| Cell Source | Autologous (patient’s own) ✓ | Often allogeneic | Mixed |
| Physician Credentials | NIH-trained MD ✓ | Varies | Varies |
Comparison is based on general regulatory frameworks. Individual clinics may vary. Japan’s MHLW certification is unique in providing government-level oversight for regenerative treatments.

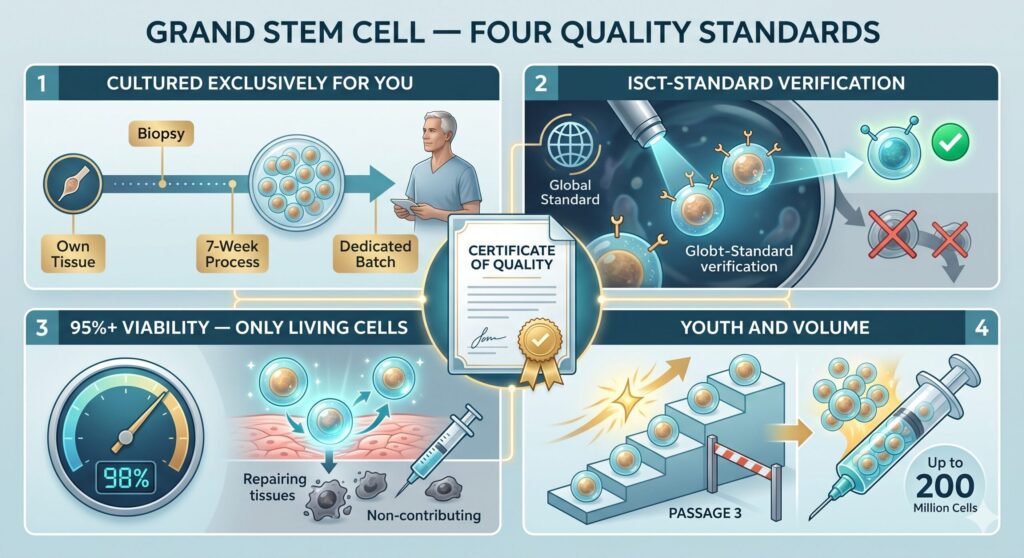
Grand Stem Cell — Four Quality Standards
Beyond the regulatory framework, Cell Grand Clinic’s cells are prepared under a four-standard quality program branded Grand Stem Cell. Each patient’s cells must meet all four before they are used.
- Cultured Exclusively for You. Grand Stem Cells are never stocked and never pooled. Cells are expanded from the patient’s own tissue over a seven-week culture process, with every batch dedicated to one patient only.
- ISCT-Standard Verification. Surface-marker testing follows the global standard of the International Society for Cell & Gene Therapy (ISCT). Cells that do not meet the defining mesenchymal stem cell criteria are discarded, not delivered.
- 95%+ Viability — Only Living Cells. Dead or degraded cells do not contribute to repair. Viability is verified at greater than 95% immediately before administration, with quality control continuing up to the moment of injection.
- Youth and Volume, Without Compromise. Stem cells age with each passage. Grand Stem Cells are strictly limited to Passage 3 or below, while scaling up to as many as 200 million cells per treatment — preserving regenerative capacity without sacrificing dose.
Every patient at Cell Grand Clinic receives a Certificate of Quality documenting that their Grand Stem Cells met all four standards before administration.
Reach us directly — WhatsApp and email inquiries are free of charge.
Frequently Asked Questions
Is stem cell therapy a real alternative to knee replacement?
Yes — for the right patient. In mild to moderate osteoarthritis, adipose-derived stem cell therapy may meaningfully alter the disease trajectory, offering durable pain relief and functional recovery that can delay or avoid the need for replacement. Even in severe cases where surgery is not an option, it remains the most effective symptom-relief choice among non-surgical treatments based on 2025 meta-analytic data.
How is stem cell therapy different from PRP?
PRP uses growth factors from your own blood platelets; stem cell therapy uses living mesenchymal stem cells, most often from your own fat. Stem cells continue releasing anti-inflammatory and repair signals for weeks, while PRP releases its growth factors over hours to days. Clinically, head-to-head data suggest stem cell therapy produces larger and longer-lasting effects than either PRP or hyaluronic acid.
Is hyaluronic acid still worth trying?
Yes, but with realistic expectations. Hyaluronic acid can reduce knee pain for weeks to months in mild osteoarthritis, and it is a low-risk procedure. In direct comparison trials, though, HA showed smaller and less durable effects than platelet- or cell-based therapies. It is a reasonable starting point, not a final answer.
When should I consider total knee replacement instead?
Total knee replacement is usually the right answer when osteoarthritis is end-stage — bone-on-bone contact, severe deformity, nighttime pain, and loss of walking capacity despite medication and physical therapy. If you can still walk reasonable distances and your imaging shows remaining cartilage, it is worth exploring regenerative options with your physician first.
What are the risks of stem cell therapy?
Autologous adipose-derived stem cell therapy has shown a favorable safety profile across more than a decade of published trials. The most common side effects are temporary injection-site swelling or mild pain. Serious adverse events have been rare in human studies when cells are processed under certified laboratory conditions — which at Cell Grand Clinic means MHLW-certified, CPC-grade processing of the patient’s own cells.
How soon will I feel a difference after stem cell treatment?
Most patients in published trials report gradual improvement over 4 to 12 weeks, with continued gains at 6 to 24 months. Stem cell therapy is not an instant pain-relief injection; it works by changing the joint environment over time. If rapid, short-term pain control is the priority, other options — including a steroid injection — may be discussed alongside stem cell therapy at Cell Grand Clinic.
Closing
Every knee is different. Your age, the grade of your arthritis, your activity goals, whether you have had previous surgery, and your general health all shape what the right answer is. For severe, end-stage osteoarthritis, total knee replacement remains a deeply effective operation — and worth the rehabilitation it demands. In earlier-stage disease, adipose-derived stem cell therapy is the one non-surgical option that aims at the underlying joint biology, with a realistic prospect of durable recovery. And when surgery is not on the table, it is still — on current evidence — the strongest symptom-relief tool we have.
If you are weighing options — or just wondering whether your knee is ready for surgery — it is reasonable to ask. Send a message to Cell Grand Clinic by WhatsApp or email. Written inquiries are free of charge, and a physician will review your imaging and history before any consultation is scheduled. For practical questions about traveling to Japan for stem cell treatment — visa, length of stay, costs, and what to expect on arrival — see our complete guide to stem cell therapy in Japan.
References
- Katz JN, Arant KR, Loeser RF. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA. 2021;325(6):568–578. https://doi.org/10.1001/jama.2020.22171
- Bennell KL, Paterson KL, Metcalf BR, et al. Effect of Intra-articular Platelet-Rich Plasma vs Placebo Injection on Pain and Medial Tibial Cartilage Volume in Patients With Knee Osteoarthritis: The RESTORE Randomized Clinical Trial. JAMA. 2021;326(20):2021–2030. https://doi.org/10.1001/jama.2021.19415
- Messier SP, Beavers DP, Queen K, et al. Effect of Diet and Exercise on Knee Pain in Patients With Osteoarthritis and Overweight or Obesity: A Randomized Clinical Trial. JAMA. 2022;328(22):2242–2251. https://doi.org/10.1001/jama.2022.21893
- Cao M, Ou Z, Sheng R, et al. Efficacy and safety of mesenchymal stem cells in knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Stem Cell Research & Therapy. 2025;16(1):122. https://doi.org/10.1186/s13287-025-04252-2
- Sun H, Zhai H, Han K, et al. Clinical outcomes of autologous adipose-derived mesenchymal stem cell combined with high tibial osteotomy for knee osteoarthritis are correlated with stem cell stemness and senescence. Journal of Translational Medicine. 2024;22(1):1039. https://doi.org/10.1186/s12967-024-05814-3
- Pers Y-M, Rackwitz L, Ferreira R, et al. Adipose Mesenchymal Stromal Cell-Based Therapy for Severe Osteoarthritis of the Knee: A Phase I Dose-Escalation Trial. Stem Cells Translational Medicine. 2016;5(7):847–856. https://doi.org/10.5966/sctm.2015-0245
- Hwang JJ, Rim YA, Nam Y, Ju JH. Recent Developments in Clinical Applications of Mesenchymal Stem Cells in the Treatment of Rheumatoid Arthritis and Osteoarthritis. Frontiers in Immunology. 2021;12:631291. https://doi.org/10.3389/fimmu.2021.631291
- Qiao X, Yan L, Feng Y, et al. Efficacy and safety of corticosteroids, hyaluronic acid, and PRP and combination therapy for knee osteoarthritis: a systematic review and network meta-analysis. BMC Musculoskeletal Disorders. 2023;24(1):926. https://doi.org/10.1186/s12891-023-06925-6
- Raeissadat SA, Ghorbani E, Sanei Taheri M, et al. MRI changes and clinical efficacy after intra-articular injection of hyaluronic acid, PRP, PRGF, and ozone in knee osteoarthritis. BMC Musculoskeletal Disorders. 2021;22(1):134. https://doi.org/10.1186/s12891-021-04017-x
- DeFrance MJ, Scuderi GR. Are 20% of Patients Actually Dissatisfied Following Total Knee Arthroplasty? A Systematic Review of the Literature. Journal of Arthroplasty. 2022;38(3):594–599. https://doi.org/10.1016/j.arth.2022.10.011
- Ma T, Jiao J, Guo D-W, et al. Incidence of periprosthetic joint infection after primary total knee arthroplasty shows significant variation: a synthesis of meta-analysis and bibliometric analysis. Journal of Orthopaedic Surgery and Research. 2024;19(1):649. https://doi.org/10.1186/s13018-024-05099-8
- Li X, Li D, Li J, et al. Preclinical Studies and Clinical Trials on Cell-Based Treatments for Meniscus Regeneration. Tissue Engineering Part B: Reviews. 2023;29(6):634–670. https://doi.org/10.1089/ten.TEB.2023.0050
- Huang L, Zhang S, Wu J, et al. Immunity-and-matrix-regulatory cells enhance cartilage regeneration for meniscus injuries: a phase I dose-escalation trial. Signal Transduction and Targeted Therapy. 2023;8(1):417. https://doi.org/10.1038/s41392-023-01670-7
Image Suggestions (for insertion during WordPress upload)
- [Image: Knee osteoarthritis infographic] — in “What Knee Osteoarthritis Really Is” — show healthy vs. worn cartilage side by side. CVR role: makes the disease mechanism tangible.
- [Image: Treatment spectrum diagram] — in “Your Full Range of Options” — horizontal gradient from “least invasive” to “most invasive”. CVR role: helps readers place themselves on the spectrum.
- [Image: ADSC harvest and injection flow] — in “Stem Cell Therapy for the Knee” — abstract clinical illustration of fat tissue → culture → knee injection. CVR role: demystifies the procedure.
- [Image: Cell Grand Clinic CPC laboratory] — in “Why Patients Travel to Japan” — sterile-environment photo of cell-processing facility with ISO cleanroom signage. CVR role: signals quality and regulatory compliance.
- [Image: Patient walking post-treatment] — in “CTA / Closing” — an active middle-aged patient walking outdoors without visible knee discomfort. CVR role: emotional anchor for the aspirational outcome.
最終更新日:2026.04.19



 Inquiries by Email
Inquiries by Email

