- Opening
- What Is Stem Cell Therapy for Erectile Dysfunction?
- Why Medication Alone Falls Short for Many Men
- How Stem Cell Therapy Restores Erectile Function
- Clinical Evidence: What the Research Shows
- Who Is a Good Candidate?
- The Treatment Process: What to Expect
- How Much Does Stem Cell Therapy for ED Cost?
- Why Japan — and Why Cell Grand Clinic?
- Frequently Asked Questions
- Take the Next Step
- References
Opening
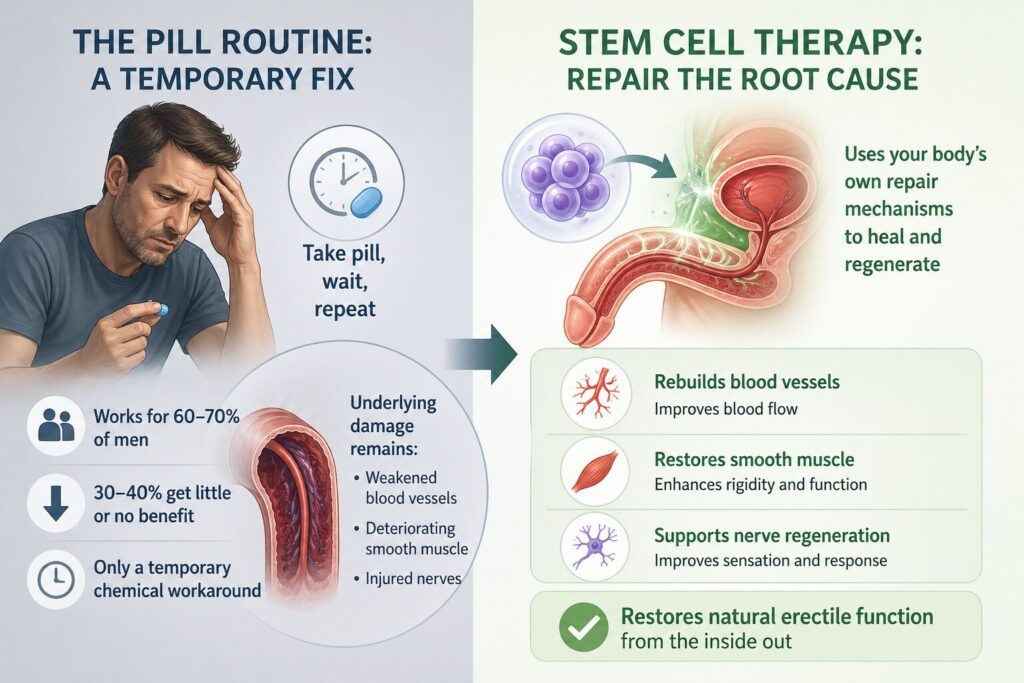
If you have been living with erectile dysfunction, you already know the routine. Take a pill, wait for it to work, hope the side effects are manageable — and repeat, every single time. For many men, this cycle becomes exhausting.
PDE5 inhibitors like Viagra and Cialis help roughly 60–70% of men — but that means 30–40% get little or no benefit at all. And even when the pills do work, they offer only a temporary chemical workaround. The moment the medication wears off, the underlying damage — weakened blood vessels, deteriorating smooth muscle, injured nerves — remains exactly where it was.
This is why a growing number of men are asking a different question: Is there a treatment that actually repairs erectile tissue, rather than masking the problem every few hours?
Stem cell therapy for erectile dysfunction is a regenerative medicine approach that uses the body’s own repair mechanisms to address the root causes of ED — rebuilding blood vessels, restoring smooth muscle, and supporting nerve regeneration. Rather than bypassing the problem with a pill, it aims to restore natural erectile function from the inside out.
What Is Stem Cell Therapy for Erectile Dysfunction?
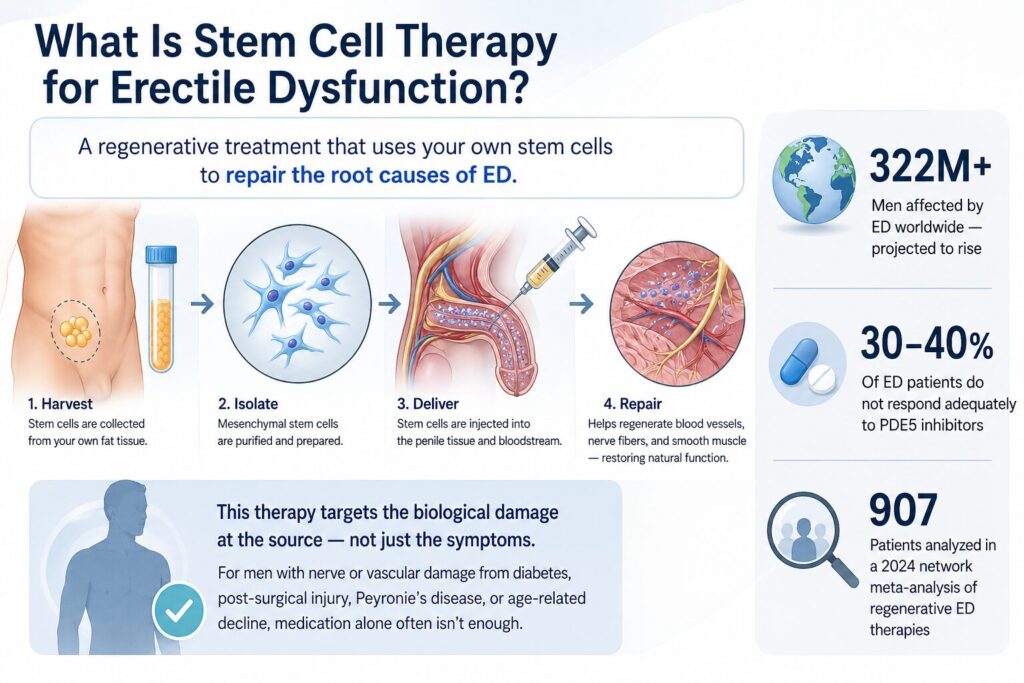
Stem cell therapy for erectile dysfunc
tion is a regenerative procedure that delivers mesenchymal stem cells — typically harvested from a patient’s own fat tissue — directly into the penile tissue and bloodstream to repair the vascular, muscular, and neural damage that causes ED.
Erectile dysfunction is far more than an inconvenience. It affects an estimated 322 million men globally, a number expected to climb as rates of diabetes, cardiovascular disease, and obesity continue to rise. While PDE5 inhibitors remain the first-line treatment, they work by temporarily blocking a single enzyme to increase blood flow for a few hours — they do not repair damaged tissue.
For men whose ED is driven by diabetes-related nerve and vascular damage, post-surgical nerve injury, Peyronie’s disease, or progressive age-related tissue degradation, medication alone often falls short. This is the gap that regenerative medicine aims to fill — not by masking the symptom, but by targeting the biological damage at its source.
Why Medication Alone Falls Short for Many Men
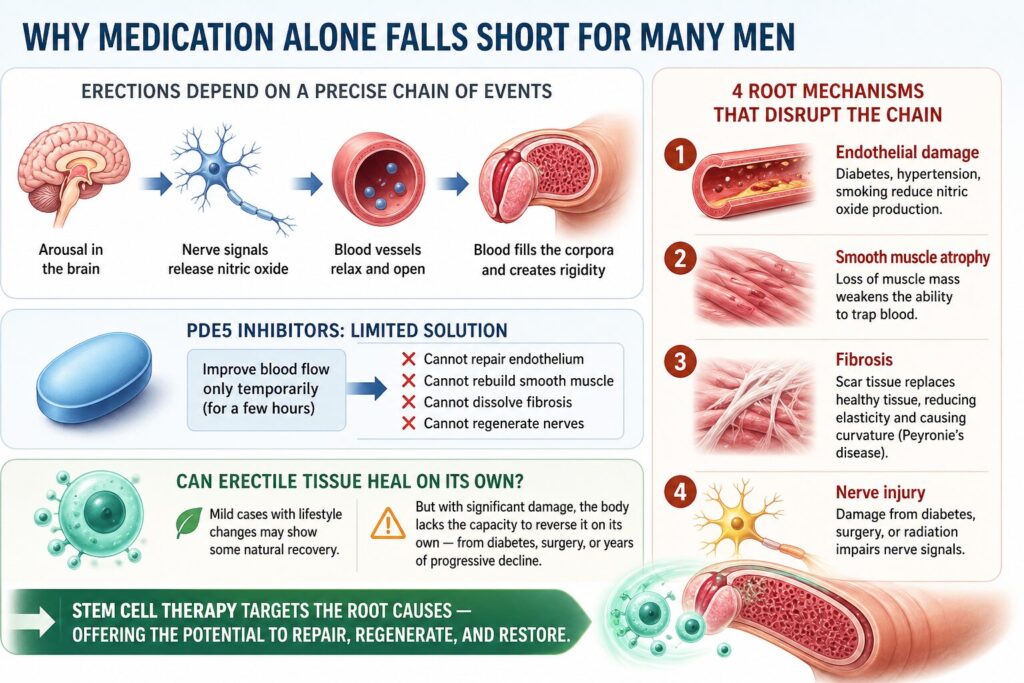
An erection requires precise coordination between the brain, nervous system, blood vessels, and penile smooth muscle. When a man becomes aroused, nerve signals trigger the release of nitric oxide, which relaxes smooth muscle in the corpora cavernosa, allowing blood to flow in and create rigidity. ED occurs when this chain is disrupted through one or more of four root mechanisms:
1. Endothelial damage — The inner lining of blood vessels deteriorates due to diabetes, hypertension, or smoking, reducing nitric oxide production.
2. Smooth muscle atrophy — The muscle within the corpora cavernosa loses mass and contractile function with age and disease, weakening the ability to trap blood.
3. Fibrosis — Collagen-rich scar tissue replaces healthy erectile tissue, reducing elasticity. In Peyronie’s disease, this causes visible curvature and functional impairment.
4. Nerve injury — The cavernous nerves that initiate erection can be damaged by diabetic neuropathy, radical prostatectomy, or pelvic radiation.
PDE5 inhibitors address only one of these four pathways — temporary blood flow enhancement — and only for a few hours at a time. They cannot repair endothelial cells, rebuild smooth muscle, dissolve fibrotic tissue, or regenerate nerves.
Can erectile tissue heal on its own? In mild cases with lifestyle changes, some limited natural recovery is possible. But when the underlying damage is significant — from diabetes, surgery, or years of progressive decline — the body lacks sufficient regenerative capacity to reverse it without intervention. This is precisely where stem cell therapy offers a fundamentally different approach.
How Stem Cell Therapy Restores Erectile Function
Mesenchymal stem cells (MSCs) restore erectile function primarily through what researchers call the paracrine effect: once delivered to damaged tissue, these cells release a powerful mix of growth factors and signaling molecules that activate the body’s own repair systems.
Unlike a medication that targets a single enzyme for a few hours, stem cells release hundreds of bioactive molecules simultaneously — addressing multiple damage pathways at once and triggering a regenerative process that continues for months after treatment.
Adipose-derived stem cells (ADSCs) — the type used in this treatment — are harvested from a patient’s own fat tissue. Fat is the richest accessible source of mesenchymal stem cells, containing approximately 500 times more MSCs per gram than bone marrow. Because the cells come from the patient’s own body, there is zero risk of immune rejection, and no immunosuppressive medication is needed.
A comprehensive 2025 review published in Frontiers in Medicine summarized the current state of stem cell research for ED, confirming that both preclinical and early clinical data support the regenerative potential of stem cells — particularly through paracrine-mediated vascular repair, smooth muscle regeneration, and nerve growth factor secretion.
Clinical Evidence: What the Research Shows
Stem cell therapy for ED is supported by a growing body of published clinical research. Most trials remain Phase I/II with relatively small sample sizes, but the consistency of positive safety signals across independent studies is notable.
| Study | Year | Design | Patients (n) | Cell Type | Key Finding | Follow-up |
|---|---|---|---|---|---|---|
| Senel et al., BMC Urol | 2025 | SR & Meta-analysis | 11 studies (6 in MA) | Mixed MSC | Significant improvement in IIEF-5, IIEF-EF, EHS, and PSV at 6 months | 6 mo |
| Hinojosa-Gonzalez et al., J Sex Med | 2024 | Network meta-analysis | 907 | Mixed (incl. SCT) | SCT SMD 0.92 (95% CrI −0.49 to 2.3); PRP and ESWT reached significance | Varied |
| Ji et al., Stem Cell Res Ther | 2025 | Prospective RCT (1:1:1) | 33 (combined arm n=11) | hPMSC + LiESWT | 70% in combined arm achieved EHS>2 at 6 months (p=0.045) | 6 mo |
| Haahr et al., EBioMedicine | 2016 | Phase I (ADRC/SVF) | 17 | Adipose-derived regenerative cells (uncultured SVF) | 47% recovered (8/17); 73% in continent men (8/11) at 6 mo | 6 mo |
| Haahr et al., Urology (12-mo FU) | 2018 | Phase I extended FU | 21 | ADRC/SVF (same as 2016) | 53% sustained recovery in continent men (8/15) at 12 mo | 12 mo |
| Nguyen Thanh et al., Stem Cell Rev Rep | 2021 | Phase I/IIa | 15 male / 31 total | Cultured autologous ADSC (IV) | All male patients reported sexual satisfaction improvement; transient testosterone increase | 12 mo |
| Al Demour et al., Urol Int | 2021 | Phase I/II | 22 | Allogeneic WJ-MSC | Significant IIEF-5, EHS, and PSV improvement over 12 mo | 12 mo |
| Yiou et al. (INSTIN), Eur Urol Focus | 2017 | Phase I/II (two-stage) | 18 (Stage I n=12, Stage II n=6) | BM-MNC | Stage II (n=6 at optimal dose): IIEF-EF 3.7→18.0 (p=0.035); Stage I 62-mo FU showed no prostate cancer recurrence | Up to 62 mo (Stage I) |
All studies used human subjects. Full references with DOI links are provided at the end of this article.
Most Recent Meta-Analysis
The most recent meta-analysis, a 2025 systematic review published in BMC Urology (Senel et al., DOI), pooled data from 11 clinical studies (6 in the meta-analysis) and found statistically significant improvements in IIEF-5, IIEF-EF, EHS, and peak systolic velocity (PSV) at six months after intracavernous stem cell injection — meaning the treatment produced measurable improvements across multiple dimensions of erectile function. The authors note that 6 months is currently the longest reported follow-up duration in this literature, and longer comparative trials are still needed.
Network Meta-Analysis of Regenerative Therapies
A 2024 Bayesian network meta-analysis published in The Journal of Sexual Medicine (Hinojosa-Gonzalez et al., DOI) compared three regenerative therapies — stem cells, platelet-rich plasma (PRP), and low-intensity shockwave therapy (LiESWT) — across 16 randomized studies involving 907 patients. Stem cell therapy had the largest point estimate for effect size (SMD 0.92), but the 95% credible interval was wide (−0.49 to 2.3) and crossed zero, meaning the result did not reach statistical significance. PRP (SMD 0.83, CrI 0.15–1.5) and shockwave therapy (SMD 0.84, CrI 0.49–1.2) both reached significance.
In plain terms: stem cells showed the numerically strongest signal but with the most uncertainty due to the smaller number of trials. PRP and shockwave therapy showed clearer statistical evidence. More large stem cell trials are needed before firm conclusions can be drawn.
Largest Systematic Review to Date
The broadest systematic review to date, published in Sexual Medicine Reviews (Furtado et al., DOI), analyzed 18 clinical trials involving 373 patients across multiple stem cell types and concluded that stem cell therapy is a promising and safe option for organic ED, while emphasizing the need for standardized techniques and larger controlled studies.
Ji 2025 — Combination Strategy in Diabetic ED
A 2025 prospective randomized controlled trial published in Stem Cell Research & Therapy (Ji et al., DOI) evaluated placenta-derived mesenchymal stem cells (hPMSCs) combined with low-intensity shockwave therapy in 33 men with refractory diabetic ED, randomized 1:1:1 to three arms. At six months, 70% of patients in the combined therapy arm (n=11) achieved an erection hardness score above 2 (p=0.045) — meaning they regained erections firm enough for sexual activity. No serious adverse events were observed.
While Cell Grand Clinic uses adipose-derived stem cells rather than placental MSCs, the broad mechanisms of MSC paracrine signaling — vascular support and nerve growth factor secretion — are studied across mesenchymal stem cell sources.
Haahr 2016 — Phase I in Post-Prostatectomy ED (ADRC / Uncultured SVF)
The Haahr et al. Phase I trial, published in EBioMedicine (2016, DOI), enrolled 17 men with severe post-prostatectomy ED who had not responded to PDE5 inhibitors. They received a single intracavernous injection of autologous adipose-derived regenerative cells (ADRCs) — also referred to in the literature as the stromal vascular fraction (SVF): a freshly isolated, uncultured cell preparation obtained directly after liposuction.
At 6 months, 8 of 17 patients (47%) recovered erectile function sufficient for intercourse without medication, and among continent patients that rate reached 73% (8 of 11).
Note on cell type: ADRC/SVF in the Haahr 2016 trial is a freshly isolated, mixed cell population (mesenchymal cells, vascular precursors, immune cells). It differs from the cultured and expanded autologous ADSCs used at Cell Grand Clinic. Both are derived from adipose tissue and share core paracrine mechanisms, but cell purity, total cell count, and regulatory categorization are different.
Haahr 2018 — 12-Month Follow-Up
A follow-up paper from the same group (Haahr et al. 2018, Urology, DOI) extended the cohort to 21 men and tracked them for 12 months. Eight of 15 continent patients (53%) retained erectile function sufficient for intercourse at one year — a meaningful sustained response, though somewhat lower than the 6-month figure. As with most regenerative therapies, individual response varies, and the modest decline at 12 months suggests that some patients may benefit from re-evaluation or repeat sessions.
Nguyen Thanh 2021 — Cultured Autologous ADSC
Among published human trials, Nguyen Thanh et al. 2021 (Stem Cell Reviews and Reports, DOI) is one of the few reports of cultured autologous ADSC used clinically for sexual function in men. In this Phase I/IIa study, 15 male participants (out of 31 total) received intravenous infusion of cultured ADSCs (1×10⁶ cells/kg) prepared at an ISO 14644-1 cell processing facility. No serious adverse events occurred over 12 months, and sexual satisfaction improved post-transplantation in all male participants. Testosterone levels rose transiently for up to 6 months. While sample size is modest, this trial is the closest published analogue to the cultured-ADSC protocol used at Cell Grand Clinic.
Al Demour 2021 — Allogeneic WJ-MSC in Diabetic ED
A Phase I/II trial of 22 diabetic ED patients, published in Urologia Internationalis (Al Demour et al. 2021, DOI), used allogeneic Wharton’s jelly-derived mesenchymal stem cells (WJ-MSCs) — donor-derived, not autologous — and demonstrated significant improvements in IIEF-5 scores, erection hardness, and penile blood flow that were sustained at 12 months. Note that this study used donor cells; Cell Grand Clinic uses autologous cells from the patient’s own tissue.
Yiou 2017 — INSTIN Long-Term Safety Data
The INSTIN trial, published in European Urology Focus (Yiou et al. 2017, DOI), was conducted in two stages. Stage I tested four doses in 12 patients, while Stage II added 6 more patients at the optimal dose (1×10⁹ BM-MNCs), for 18 total participants.
In Stage II patients (n=6), six-month outcomes showed IIEF-EF score improvement from 3.7 to 18.0 (p=0.035) — a marked increase in erectile function score. In Stage I patients (n=12), long-term follow-up extended to a mean of 62 months, and no prostate cancer recurrences were observed — supporting the long-term safety of intracavernous cell therapy. Erectile function scores in Stage I declined modestly from the 1-year peak, suggesting that repeat sessions may sometimes be appropriate.
Safety across all trials: Published clinical studies consistently report no serious adverse events from intracavernous stem cell injection for ED. Side effects have been limited to mild, temporary swelling or bruising at the injection site, resolving within days.
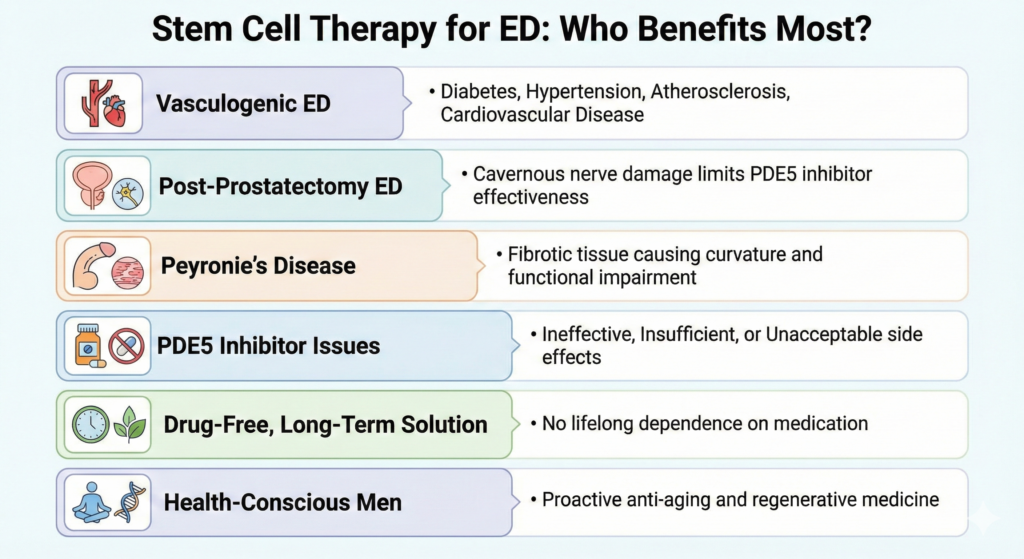
Who Is a Good Candidate?
Stem cell therapy for erectile dysfunction may be particularly beneficial for men whose ED has not responded adequately to conventional treatment, or who seek a drug-free alternative that addresses the underlying cause.
Individual suitability is determined during a medical consultation. This checklist is for general reference only.
Reach us directly — WhatsApp and email inquiries are free of charge.
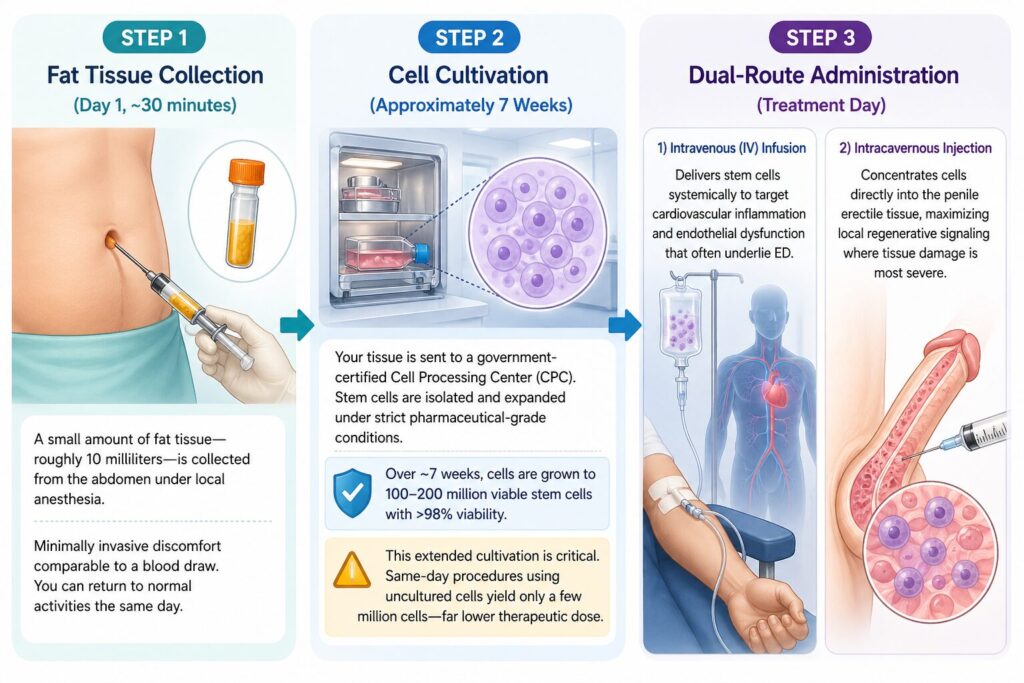
The Treatment Process: What to Expect
Step 1 — Fat Tissue Collection (Day 1, approximately 30 minutes)
A small amount of fat tissue — roughly 10 milliliters — is collected from the abdomen under local anesthesia. The procedure is minimally invasive and comparable in discomfort to a standard blood draw. You can return to normal activities the same day.
On the very same day as your fat collection, you also have the option to receive exosome therapy. Culturing your own stem cells takes several weeks, and many patients don’t want to simply wait while their symptoms continue. Exosomes—the tiny signaling messengers naturally released by stem cells—can help calm inflammation and ease discomfort during this period, so you’re already starting to feel better by the time your cultured cells are ready. Think of it as a bridge: exosomes work with your treatment timeline, not against it.
Step 2 — Cell Cultivation (Approximately 7 Weeks)
Your harvested tissue is sent to a government-certified Cell Processing Center (CPC), where stem cells are isolated and expanded under strict pharmaceutical-grade conditions. Over approximately seven weeks, cells are grown to reach a therapeutic dose of 100 to 200 million viable stem cells with a verified viability rate above 98%.
This extended cultivation period is critical. Some clinics offer same-day procedures using uncultured cells — often yielding only a few million cells. The difference in therapeutic dose is substantial.
Step 3 — Dual-Route Administration (Treatment Day)
Intravenous (IV) infusion delivers stem cells systemically, targeting the cardiovascular inflammation and endothelial dysfunction that often underlie ED. Intracavernous injection concentrates cells directly into the penile erectile tissue, maximizing local regenerative signaling where tissue damage is most severe.
Recovery Timeline
Weeks 1–4: Stem cells engraft and begin releasing paracrine factors. Many patients notice improved energy and circulation.
Months 1–3: New blood vessel formation begins. Some patients experience improved firmness, increased sensitivity, or the return of morning erections.
Months 3–6: Peak improvement period. Vascular maturation, smooth muscle proliferation, and nerve pathway restoration. Many patients report significantly improved erectile quality and return of spontaneous function.
Months 6–12+: Published studies show improvements tend to be durable, maintained at 12-month follow-up — because stem cells promote structural tissue repair rather than pharmacological dependence.
If you are exploring stem cell therapy for diabetes or anti-aging treatments alongside ED, these conditions can often be addressed within the same treatment protocol, as the systemic IV component benefits multiple organ systems simultaneously.
Reverse Aging: How Stem Cell IV Therapy Actually Works Learn How Stem Cell Therapy for Diabetes: A Regenerative Approach to Glycemic Control Learn More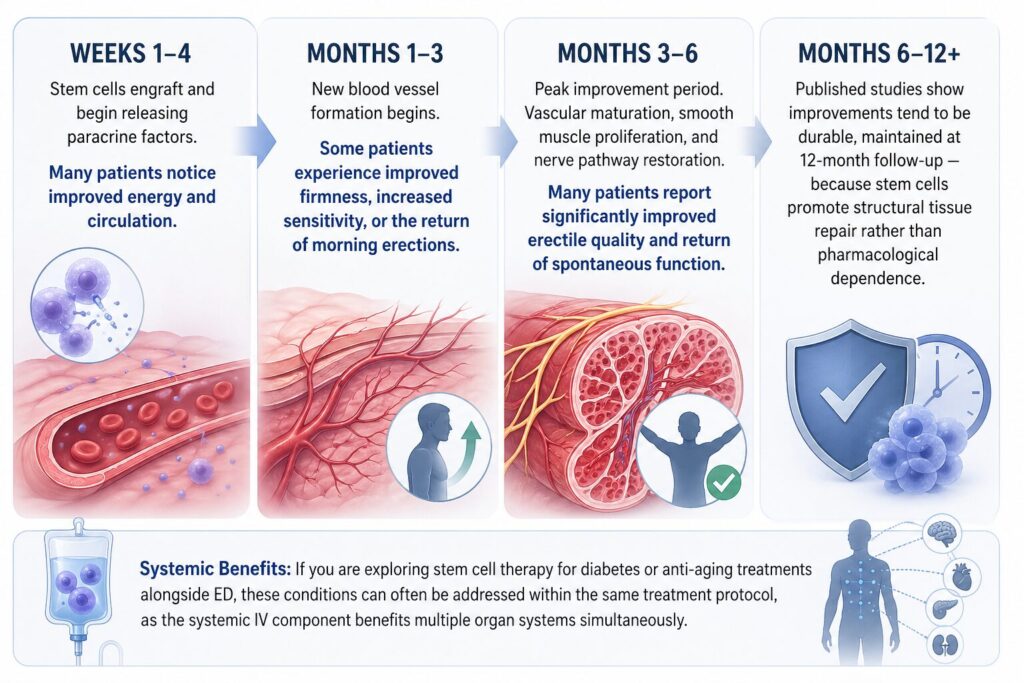
How Much Does Stem Cell Therapy for ED Cost?
Stem cell therapy for ED is not covered by health insurance in any country, as it is classified as an advanced regenerative treatment. Costs vary significantly depending on the clinic, cell quality, and treatment protocol.
In Japan, high-quality autologous stem cell therapy using cultured, expanded cells typically ranges from approximately $15,000 to $50,000 USD per session. The variation reflects differences in cell count, cultivation duration, quality assurance protocols, and the number of administration routes.
Why the wide range? Think of it this way: just as the production process behind A5 Wagyu beef differs fundamentally from a standard cut — not merely in taste but in every stage of care and quality control — a 7-week, 200-million-cell, dual-route protocol with full quality certification differs fundamentally from a same-day procedure using a fraction of the cells with minimal oversight.
When comparing costs internationally, consider what is included: cell processing center certification, cell viability verification, quality certificates for every batch, government regulatory oversight, and physician expertise. The cheapest option is rarely the safest or most effective.
Specific pricing varies by individual case. Contact Cell Grand Clinic directly for a personalized assessment.
Cell Quality: The Key Factor That Impacts Stem Cell Therapy Outcomes Learn MoreWhy Japan — and Why Cell Grand Clinic?
Japan’s Regenerative Medicine Regulatory Framework
Japan is one of the only countries with a comprehensive legal framework specifically designed for regenerative medicine. The Act on the Safety of Regenerative Medicine (enacted 2013, amended 2020) requires every stem cell treatment plan to be reviewed and approved by an independent MHLW-Certified Special Committee before it can be offered to patients.
This is not self-regulation — it is government-mandated oversight with real accountability. Every treatment protocol, every cell processing step, and every safety measure is documented and monitored.
| Factor | Japan (MHLW-certified) | Mexico | Thailand |
|---|---|---|---|
| Government Oversight | MHLW certified ✓ | Limited regulation | Emerging regulation |
| Cell Quality Standards | CPC-grade, 7-week culture ✓ | Varies by clinic | Varies by clinic |
| Cell Count per Treatment | Up to 200 million ✓ | Varies widely | Varies widely |
| Autologous (Own Cells) | Yes ✓ | Often allogeneic | Mixed |
| Physician Credentials | NIH-trained, ABRM Diplomate ✓ | Varies | Varies |
Comparison is based on general regulatory frameworks. Individual clinics may vary. Japan’s MHLW certification system (Act on the Safety of Regenerative Medicine) is unique in providing government-level oversight for regenerative treatments.
Cell Grand Clinic: What Sets Us Apart
Cell Grand Clinic operates under Japan’s Act on the Safety of Regenerative Medicine, holding 13 government-approved treatment plans — among the highest numbers of any regenerative medicine clinic in Japan (Plan No. PB5240089 and others).

The clinic is directed by Dr. Yuichi Wakabayashi, M.D., Ph.D., a Diplomate of the American Board of Regenerative Medicine (ABRM) with research experience at the U.S. National Institutes of Health (NIH). With over 3,000 stem cell treatments performed, Dr. Wakabayashi brings both international research credentials and deep clinical experience to every patient’s treatment plan.
Cell quality is central to our approach: every batch is cultivated for approximately 7 weeks at a certified CPC, tested for viability (98%+), screened for infectious agents, and accompanied by a certificate of quality. We never use frozen, pre-made, or off-the-shelf cell products.
Cell Quality: The Key Factor That Impacts Stem Cell Therapy Outcomes Learn MoreFrequently Asked Questions
Can stem cells repair erectile dysfunction?
Early clinical evidence suggests stem cell therapy may improve erectile function by supporting repair of damaged blood vessels, smooth muscle, and nerves. A 2025 meta-analysis (Senel et al.) found significant improvements in erectile function scores at six months. However, results vary by individual, and stem cell therapy for ED is still classified as an emerging treatment. Larger trials are underway to establish long-term efficacy.
How much does stem cell therapy for erectile dysfunction cost?
In Japan, autologous stem cell therapy for ED typically costs approximately $15,000–$50,000 USD per session, depending on cell count, cultivation protocol, and administration method. This is not covered by insurance. Pricing varies by individual case — contact Cell Grand Clinic for a personalized estimate.
How long does stem cell therapy for ED take to work?
Most patients begin noticing initial changes within one to three months, with peak effects in published studies typically occurring between three and six months after treatment. Because stem cells work through tissue support rather than pharmacological effect, the process is gradual. Published studies show benefits often maintained at 12-month follow-up (e.g., Haahr 2018: 53% sustained recovery), though individual durability varies.
Is stem cell therapy for ED safe?
Across published clinical trials of intracavernous stem cell injection for ED, no serious adverse events have been reported. Side effects are limited to mild, temporary swelling or bruising at the injection site. Because Cell Grand Clinic uses autologous (your own) cells, there is no risk of allogeneic immune rejection.
What is the success rate of stem cell therapy for ED?
Success rates vary by study and patient population. In the Haahr et al. 2016 trial (using freshly isolated ADRC/SVF), 47% of men with severe post-prostatectomy ED recovered function after a single injection at 6 months — and 73% among continent patients. The 12-month follow-up (Haahr 2018) showed 53% sustained recovery in continent patients. In the INSTIN trial Stage II (n=6), IIEF-EF scores improved from 3.7 to 18.0 at six months. A 2023 systematic review of 18 clinical studies involving 373 patients found consistent safety and efficacy signals across multiple stem cell types. Individual results depend on the cause and severity of ED, and outcomes cannot be predicted in advance.
Can stem cells cure erectile dysfunction permanently?
No treatment can guarantee a permanent cure, and it would be inaccurate to claim otherwise. Stem cell therapy aims to produce durable improvement by supporting underlying tissue repair, but durability varies between patients. Published trials show some sustained benefits at 12+ months in responders, while some patients may require additional sessions.
Can erectile tissue heal on its own?
In mild cases, lifestyle changes such as improved diet, exercise, and managing conditions like diabetes can support some natural recovery. However, when significant structural damage exists — from disease, surgery, or years of progressive decline — the body’s natural repair capacity is often insufficient. Stem cell therapy may provide additional regenerative support beyond what damaged erectile tissue can generate on its own.
Can stem cells increase size?
Stem cell therapy for ED is designed to support natural erectile function — not to increase penile size. The treatment works by supporting tissue repair and blood flow, which may result in firmer, fuller erections closer to a patient’s natural baseline. Claims about permanent size increase from stem cells are not supported by current clinical evidence.
Is stem cell therapy for ED approved by the FDA or Japan’s MHLW?
In the United States, the FDA has not approved stem cell therapy for ED. In Japan, stem cell therapy operates under a different legal framework: the Act on the Safety of Regenerative Medicine, which requires government-certified committee review of every treatment plan. Cell Grand Clinic’s treatment plans have passed this review process. This is distinct from “FDA approval” — it provides regulated safety oversight, not an efficacy guarantee.
How does stem cell therapy compare to PRP for erectile dysfunction?
A 2024 network meta-analysis in The Journal of Sexual Medicine (Hinojosa-Gonzalez et al.) compared stem cells, PRP, and shockwave therapy across 907 patients. Stem cells showed the largest point estimate (SMD 0.92), but the 95% credible interval was wide and crossed zero, meaning the effect did not reach statistical significance in this analysis. PRP (SMD 0.83) and shockwave therapy (SMD 0.84) both reached statistical significance. Stem cells showed the numerically strongest signal but with greater uncertainty due to fewer trials. The optimal choice depends on individual clinical factors.
Does stem cell therapy work for ED after prostatectomy?
The Haahr et al. 2016 Phase I trial enrolled 17 men with post-prostatectomy ED who had failed PDE5 inhibitor treatment, using freshly isolated adipose-derived regenerative cells (ADRC/SVF). At 6 months, 73% of continent patients (8 of 11) recovered erectile function sufficient for intercourse. The 12-month follow-up (Haahr 2018, n=21) reported 53% sustained recovery in continent patients. Cavernous nerve repair support is one of the proposed mechanisms by which stem cell therapy may help recovery after prostate surgery, though individual results vary.
Take the Next Step
Erectile dysfunction is not something you simply have to accept. It is a medical condition with identifiable biological causes — and regenerative medicine offers a genuine pathway to address those causes rather than masking them with medication.
If PDE5 inhibitors have not worked for you, if your ED is driven by diabetes, surgery, or progressive vascular disease, or if you want an evidence-based, drug-free approach to restoring your sexual health — we invite you to explore what stem cell therapy at Cell Grand Clinic can offer.
Reach us directly — WhatsApp and email inquiries are free of charge.
References
Senel S, et al. Stem cell therapy for erectile dysfunction: promise or reality? — a systematic review and meta-analysis of clinical trials. BMC Urol. 2025;25(1):222. https://doi.org/10.1186/s12894-025-01913-5
Hinojosa-Gonzalez DE, et al. Regenerative therapies for erectile dysfunction: a systematic review, Bayesian network meta-analysis, and meta-regression. J Sex Med. 2024;21(12):1152-1158. https://doi.org/10.1093/jsxmed/qdae131
Furtado TP, et al. Stem cell therapy for erectile dysfunction: a systematic review. Sex Med Rev. 2023;12(1):87-93. https://doi.org/10.1093/sxmrev/qead040
Ji YH, et al. High-activity placenta-derived mesenchymal stem cells combined with low-intensity extracorporeal shock wave therapy for diabetic erectile dysfunction: a prospective randomized controlled trial. Stem Cell Res Ther. 2025;16(1):359. https://doi.org/10.1186/s13287-025-04499-9
Haahr MK, et al. Safety and potential effect of a single intracavernous injection of autologous adipose-derived regenerative cells in patients with erectile dysfunction following radical prostatectomy: an open-label phase I clinical trial. EBioMedicine. 2016;5:204-210. https://doi.org/10.1016/j.ebiom.2016.01.024
Nguyen Thanh L, et al. Can autologous adipose-derived mesenchymal stem cell transplantation improve sexual function in people with sexual functional deficiency? Stem Cell Rev Rep. 2021;17(6):2153-2163. DOI
Al Demour S, et al. Safety and efficacy of 2 intracavernous injections of allogeneic Wharton’s jelly-derived mesenchymal stem cells in diabetic patients with erectile dysfunction: Phase 1/2 clinical trial. Urol Int. 2021;105(11-12):935-943. https://doi.org/10.1159/000517364
Yiou R, et al. Intracavernous injections of bone marrow mononucleated cells for postradical prostatectomy erectile dysfunction: final results of the INSTIN clinical trial. Eur Urol Focus. 2017;3(6):643-645. https://doi.org/10.1016/j.euf.2017.06.009
Fu X, et al. Advances in stem cell therapy for erectile dysfunction: preclinical evidence and emerging therapeutic approaches. Front Med. 2025;12:1519095. https://doi.org/10.3389/fmed.2025.1519095
最終更新日:2026.04.28



 Inquiries by Email
Inquiries by Email

