Stem cell therapy for diabetes uses adipose-derived mesenchymal stem cells to regenerate insulin-producing beta cells, reverse insulin resistance, and repair complications — for patients whose medication keeps climbing while neuropathy, nephropathy, or foot ulcers advance.
Below: how stem cells regenerate beta-cell function, what the 2025 meta-analysis of 34 RCTs reports, and why Japan’s MHLW-certified framework gives outcomes you can verify.
At Cell Grand Clinic in Osaka, Japan, treatment delivers up to 200 million autologous stem cells via IV — cultured 7 weeks from your own minimal fat tissue. Never donor cells.
MHLW Type 2 certified. NIHtrained physician. 3,000+ stem cell cases. Diabetes Plan No. PB5250050.
- What Is Stem Cell Therapy for Diabetes?
- Why Conventional Therapy Hits a Wall
- How Adipose-Derived Stem Cells Treat Diabetes
- Type 1 vs Type 2 Diabetes Stem Cell Treatment: Which Responds Best?
- Beyond Blood Sugar: Treating Diabetic Complications with Stem Cells
- Stem Cell Therapy for Diabetes: Success Rate and Clinical Evidence
- Is Stem Cell Therapy Right for Your Diabetes?
- Where Can You Get Stem Cell Treatment for Diabetes? Why Japan?
- Why Cell Grand Clinic for Diabetes Stem Cell
- The Treatment Protocol for International Patients
- What to Expect: Timeline of Stem Cell Therapy Results
- Frequently Asked Questions
- Take Control: From Symptom Management to Regeneration
What Is Stem Cell Therapy for Diabetes?
Stem cell therapy for diabetes is a regenerative medicine approach that uses mesenchymal stem cells to regenerate insulin-producing beta cells, reverse insulin resistance, and repair diabetic complications including neuropathy, nephropathy, and retinopathy. A 2025 meta-analysis of 34 randomized controlled trials found that stem cell therapy is 2.79 times more effective than placebo at improving glycemic control (OR 2.79, 95% CI 1.63–4.75).
Stem cell therapy is not a fantasy—it’s a clinical reality refined across hundreds of peer-reviewed studies. And in Japan, it’s regulated, accessible, and increasingly proven for patients with type 1, type 2, and diabetic complications that conventional medicine cannot repair.
Why Conventional Therapy Hits a Wall
Insulin and oral medications control blood sugar but do not regenerate exhausted beta cells,
reverse insulin resistance, or repair the microvascular damage driving long-term complications.
More than 50% of long-term diabetes patients develop neuropathy, nephropathy, retinopathy, or
cardiovascular disease — outcomes that medication alone cannot prevent or reverse.

You take your medication every morning. You monitor your blood sugar. You watch your diet and follow every recommendation your doctor gives you.
And still — the neuropathy in your feet keeps spreading. Your nephrologist has started mentioning dialysis. Your ophthalmologist talks about laser treatment. The medication doses keep climbing. Somewhere in the back of your mind is a fear you rarely say out loud: that you are doing everything right, and the disease is still winning.
This experience — the quiet frustration of conscientious self-management against a disease that keeps advancing — is shared by hundreds of millions of people worldwide. Over 537 million people worldwide have diabetes, yet current insulin-based and oral medication strategies address only the symptoms—high blood sugar. They do not regenerate pancreatic beta cells. They do not reverse insulin resistance in muscle and liver. And they do not repair the microvascular and neuropathic damage that accumulates silently over years.
How Adipose-Derived Stem Cells Treat Diabetes
Adipose-derived stem cells (ADSCs) act through four mechanisms: they release growth factors
that protect and regenerate pancreatic beta cells, restore GLUT4 and IRS-1/AKT insulin signaling
pathways, suppress chronic inflammation via M1-to-M2 macrophage conversion, and improve
liver glucose metabolism. This systemic action targets the underlying disease, not only its
symptoms.

β-Cell Regeneration and Protection
Stem cells release growth factors — including VEGF, TGF-β, IGF-1, and HGF — that stimulate pancreatic β-cell regeneration and protect existing β-cells from further destruction. They reduce oxidative stress and suppress inflammatory cell death pathways.
For Type 1 Diabetes specifically, ADSCs modulate the misdirected autoimmune response that destroys β-cells — addressing the root immunological cause rather than compensating for its downstream effect. For Type 2 Diabetes, the focus shifts to regenerating β-cells exhausted by years of overdemand.
Reversal of Insulin Resistance at the Cellular Level
ADSC therapy increases GLUT4 expression — the glucose transporters in liver, muscle, and adipose tissue through which insulin moves glucose into cells. It restores the IRS-1/AKT signaling pathway, the primary molecular route through which insulin communicates with target tissues. It suppresses the NLRP3 inflammasome, a key inflammatory trigger that blocks insulin signaling.
This is not modest improvement in insulin sensitivity through a single pathway. This is restoration of the cellular machinery that makes insulin function properly — the same machinery that years of insulin resistance have progressively degraded.
Liver Function Restoration
The liver plays a central role in glucose regulation, and hepatic dysfunction is a significant contributor to both fasting and post-meal blood sugar elevation in diabetes. ADSC therapy improves liver function markers, reduces hepatic fat accumulation, and promotes glycogen synthesis — directly improving glucose metabolism through a route that oral medications cannot access.
Systemic Anti-Inflammatory Reset
Chronic low-grade inflammation is now recognized as a primary driver of both Type 1 and Type 2 Diabetes, not merely a consequence of them. ADSCs convert pro-inflammatory M1 macrophages to anti-inflammatory M2 macrophages, expand regulatory T cells for sustained immune balance, and shift the systemic cytokine profile from destructive to protective.
No single pharmaceutical achieves this breadth of anti-inflammatory action. It is a system-wide reset — and it is the same mechanism through which ADSC therapy also addresses diabetic complications.
Type 1 vs Type 2 Diabetes Stem Cell Treatment: Which Responds Best?
Stem cell treatment works for both Type 1 and Type 2 diabetes through different mechanisms. In
Type 1, stem cells suppress the autoimmune attack destroying beta cells. In Type 2, they reverse
insulin resistance and regenerate exhausted beta cells. The 2025 meta-analysis reported an odds
ratio of 2,56 for T1DM and 6.76 for T2DM compared to placebo.
| Factor | Type 1 Diabetes | Type 2 Diabetes |
|---|---|---|
| Root cause | Autoimmune destruction of β-cells | Insulin resistance + β-cell exhaustion |
| Primary stem cell action | Immune modulation — suppresses the autoimmune attack that destroys β-cells | Metabolic restoration — reverses insulin resistance and regenerates exhausted β-cells |
| Key molecular pathway | Regulatory T-cell expansion, pro-inflammatory cytokine suppression | GLUT4 upregulation, IRS-1/AKT restoration, NLRP3 inflammasome suppression |
| Clinical evidence strength | Pilot-stage — phase 1b/2a (Araujo et al., 2020: 2 of 8 achieved insulin independence) | Strong — meta-analytic (Li et al., 2025: OR 6.76 for T2DM specifically) |
| Best timing | Within 1–2 years of diagnosis (while β-cells remain) | Any stage — earlier treatment yields stronger results |
| Complication treatment | Yes — neuropathy, nephropathy, retinopathy | Yes — full range including foot ulcers, ED, cardiovascular |
Beyond Blood Sugar: Treating Diabetic Complications with Stem Cells
Stem cell therapy directly targets the diabetic complications that drug therapy cannot address:
neuropathy, nephropathy, retinopathy, foot ulcers, erectile dysfunction, and cardiovascular
disease. By restoring microcirculation, regenerating nerves, and reducing inflammation, ADSC
therapy works on the underlying tissue damage — keeping limbs, vision, and kidney function that
conventional treatment can only slow.
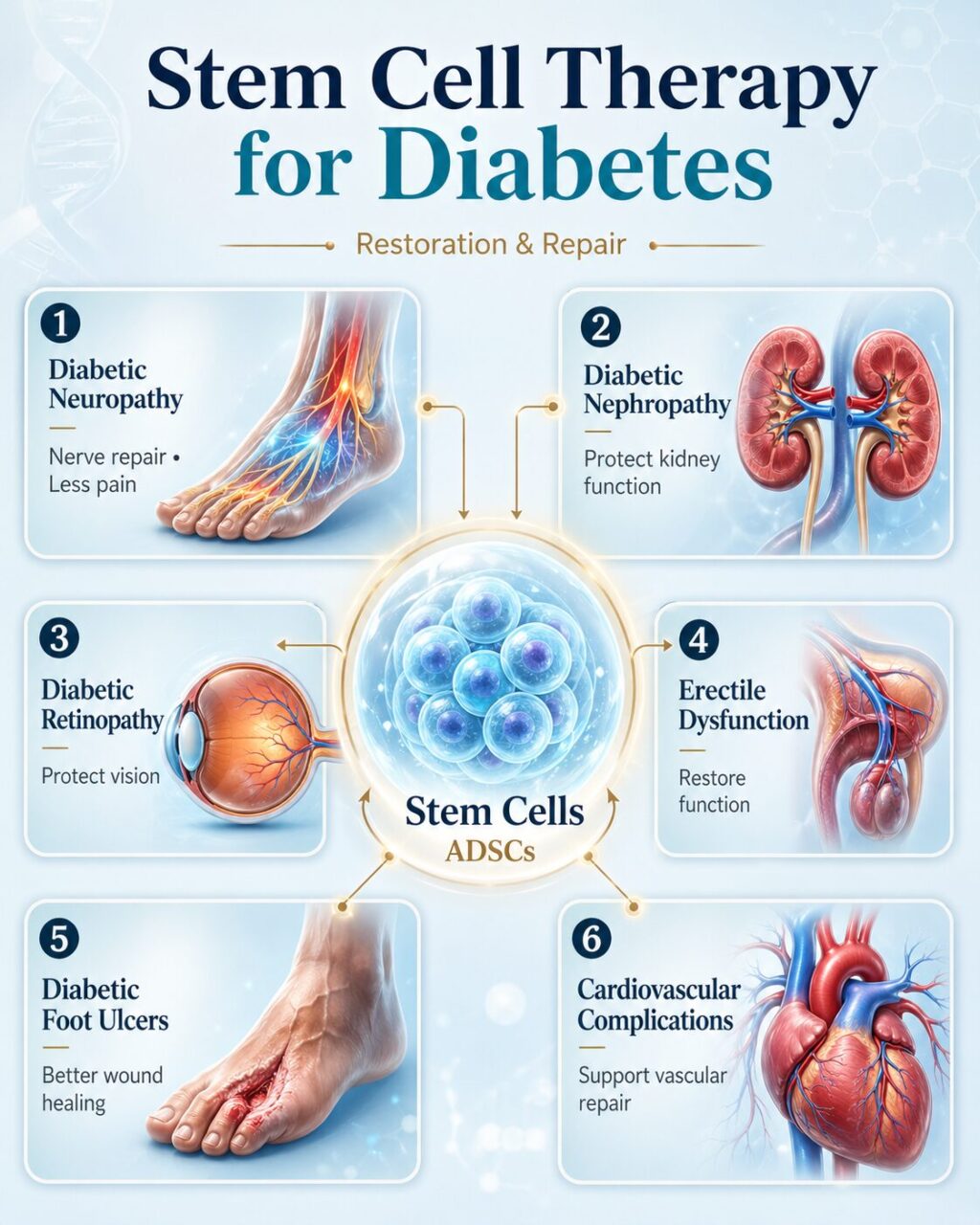
Diabetic Neuropathy
Neuropathic pain diminishes as stem cells regenerate nerve endings and restore blood flow. Many patients report improved sensation, warmth, and reduced numbness within 3–6 months.
Diabetic Nephropathy (Kidney Disease)
Kidney function stabilizes as stem cells reduce glomerular inflammation and promote podocyte repair. eGFR often improves in patients with early-stage diabetic nephropathy.
Diabetic Retinopathy
Vision and retinal health improve through restored retinal blood flow and reduced inflammation.
Diabetic Foot Ulcers
Enhanced wound healing through tissue regeneration and new blood vessel formation — directly addressing the condition that leads to amputation. Diabetic foot ulcers are the leading cause of non-traumatic lower limb amputation — this is the biological pathway that regenerative medicine is now demonstrating the ability to interrupt.
Erectile Dysfunction
Improved microcirculation and nerve function restores natural function — a result that PDE5 inhibitors (like Viagra) can only temporarily simulate. The mechanism and clinical evidence for this application are covered in depth in our article on stem cell therapy for erectile dysfunction.
Cardiovascular Complications
Improved endothelial function and reduced vascular inflammation — the most common cause of death in diabetic patients.
Reach us directly — WhatsApp and email inquiries are free of charge.
Stem Cell Therapy for Diabetes: Success Rate and Clinical Evidence
Stem cell therapy for diabetes has a success rate approximately 3 times higher than placebo for improving glycemic control. A 2025 meta-analysis of 34 randomized controlled trials (Li et al., Stem Cell Research & Therapy) reported an odds ratio of 2.79 (95% CI 1.63–4.75) — meaning patients who received stem cell therapy were nearly 3 times more likely to achieve meaningful improvement.
| Study Focus | Study Type | Key Finding | What It Means |
|---|---|---|---|
| Overall Glycemic Control | Meta-analysis (34 RCTs) | OR 2.79 | Nearly 3× more likely to improve blood sugar control |
| Autologous MSCs Specifically | Subgroup analysis | OR 5.28 | Using your own cells = nearly 5× more effective |
| Diabetic Foot Ulcers | Systematic review | RR 1.56 | 56% more likely to heal — wounds closed ~19 days faster |
| Diabetic Kidney Disease | Meta-analysis (4 trials, n=90) | eGFR improved (P=0.02) | Kidney filtration improved — usually considered irreversible |
| Type 1 Diabetes | Phase 1b/2a (n=13) | 2 of 8 insulin-free | 2 patients stopped insulin entirely in early onset T1DM; most reduced dosage |
Is Stem Cell Therapy Right for Your Diabetes?
You may benefit from stem cell therapy if you have Type 2 diabetes inadequately controlled by
medication, diabetic complications such as neuropathy or kidney disease, foot ulcers resistant to
standard care, or recently-diagnosed Type 1 diabetes (within 1–2 years). Active infection, current
cancer, and severe organ failure remain absolute contraindications.
- Type 2 diabetes not fully controlled by medication
- Diabetic neuropathy (numbness, tingling, pain in hands/feet)
- Early-stage diabetic kidney disease (declining eGFR)
- Diabetic foot ulcers resistant to standard wound care
- Diabetic erectile dysfunction
- Recent-onset type 1 diabetes (within 1–2 years of diagnosis)
- Active infection or sepsis
- Recent or active cancer
- Severe organ failure (end-stage kidney or liver)
- Inability to commit to follow-up monitoring
Where Can You Get Stem Cell Treatment for Diabetes? Why Japan?
Stem cell treatment for diabetes is available in Japan, Mexico, Thailand, Germany, and Panama,
but regulatory rigor varies enormously. Japan is the only country with a dedicated national law —
the Act on the Safety of Regenerative Medicine (2014) — requiring government-registered
protocols, MHLW-certified cell processing, and ongoing safety monitoring for every treatment.
Japan’s Regenerative Medicine Regulatory Framework
Japan operates under the world’s most stringent regenerative medicine oversight: the Ministry of Health, Labour and Welfare (MHLW). Unlike offshore clinics in Mexico or Thailand, every stem cell treatment in Japan is registered, tracked, and regularly audited.
This means no corner-cutting, no unproven protocols, no commercialism overriding patient safety. Treatment protocols must demonstrate safety before approval. Your treatment is logged into a national regenerative medicine registry.
| Factor | 🇯🇵 Japan | 🇲🇽 Mexico | 🇹🇭 Thailand | 🇩🇪 Germany |
| Dedicated stem cell law | ✅ Yes (2014) | ❌ None | ❌ None | 🟡 Partial |
| Gov. committee review required | ✅ Mandatory | ❌ No | ❌ No | 🟡 Varies |
| Licensed CPC (GMP-standard) | ✅ Mandatory | 🟡 Variable | 🟡 Variable | ✅ Yes |
| National adverse event database | ✅ Mandatory | ❌ No | ❌ No | 🟡 Partial |
| Autologous ADSC legal status | ✅ Certified & regulated | 🟡 Gray zone | 🟡 Gray zone | 🟡 Limited |
| Regulatory transparency | ✅ High | 🔴 Low | 🔴 Low | ✅ High |
Why Cell Grand Clinic for Diabetes Stem Cell
Cell quality: Fresh, Pure, and Young.
Cells are expanded at a government-certified Cell Processing Center over approximately 7 weeks — a culture duration deliberately maintained to reach therapeutic concentrations of up to 200 million cells per session. Each batch is verified for viability (≥95%), purity via multi-marker surface antigen screening (CD73+, CD90+, CD105+; CD45–, CD34–), and low passage (P3) before administration. Low-passage cells retain full regenerative capacity; clinics that rush culture timelines or use high-passage cells deliver a biologically diminished product.

13 MHLW-certified treatments
The clinic holds government-certified Regenerative Medicine Provision Plans under Japan’s Act on the Safety of Regenerative Medicine for 13 distinct treatments — including the specific Type 2 plan for Diabetes Mellitus and Prediabetes (Plan No. PB5250050). This is not a general operating license; each individual treatment plan requires independent government committee review and approval.

Physician credentials
The Medical Director trained at the U.S. National Institutes of Health (NIH) in neurological disorders research and holds Diplomate status with the American Board of Regenerative Medicine (ABRM) — the primary international board certification in this specialty. Over 3,000 stem cell treatments have been performed, spanning osteoarthritis, diabetes, chronic pain, frailty, erectile dysfunction, hair loss, and aesthetic rejuvenation. Research achievements include first-author publication of the world’s first-in-human use of a PDE4B-specific PET tracer, conducted in collaboration with Pfizer Inc., and media coverage in The Wall Street Journal.
The Treatment Protocol for International Patients
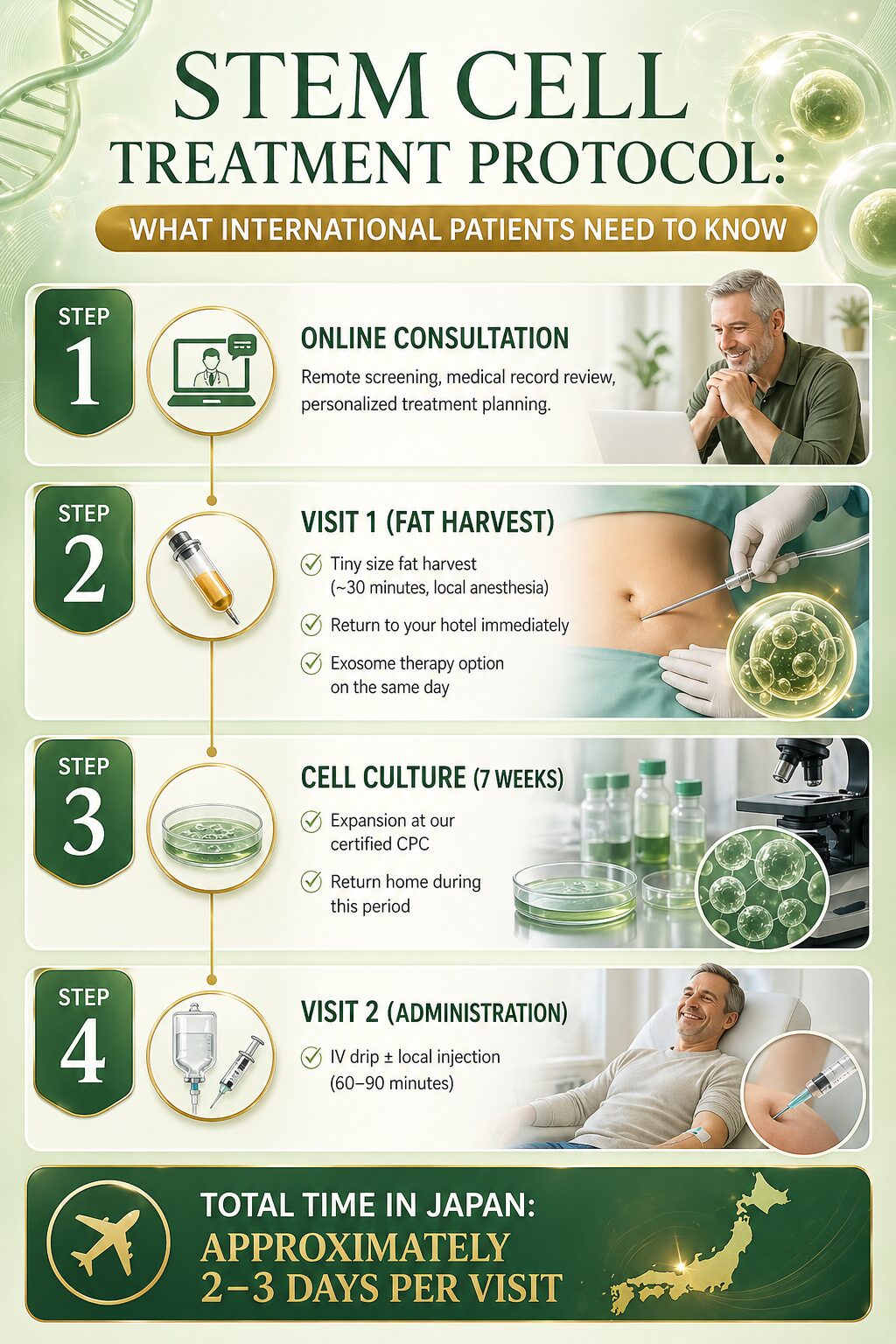
Step 1 — Online Consultation: Remote screening, medical record review, personalized treatment planning.
Step 2 — Visit 1 (Fat Harvest): Tiny size fat harvest (~30 minutes, local anesthesia). Return to your hotel immediately.
On the very same day as your fat collection, you also have the option to receive exosome therapy. Culturing your own stem cells takes several weeks, and many patients don’t want to simply wait while their symptoms continue. Exosomes—the tiny signaling messengers naturally released by stem cells—can help calm inflammation and ease discomfort during this period, so you’re already starting to feel better by the time your cultured cells are ready. Think of it as a bridge: exosomes work with your treatment timeline, not against it.
Step 3 — Cell Culture (7 weeks): Expansion at our certified CPC. Return home during this period.
Step 4 — Visit 2 (Administration): IV drip ± local injection (60–90 minutes).
Total time in Japan: approximately 2–3 days per visit.
Innovative W-injection ~Our feature~
Systemic IV drip: 100–200 million ADSCs administered intravenously over 60–90 minutes. This is the primary route for Type 1 and Type 2 Diabetes, delivering cells systemically to address pancreatic function, insulin resistance, and vascular inflammation throughout the body.
Local injection: Customized dosing for targeted complications such as diabetic erectile dysfunction, where cavernosal injection achieves higher local cell concentration at the tissue level.
Combination protocol: IV drip plus local injection for patients with both systemic disease and specific complication targets.
Drug-Free ED Treatment: Stem Cells Without Pills or Implants Learn More Exosome Therapy: Same-Day, Cell-Free Regeneration Learn MoreWhat to Expect: Timeline of Stem Cell Therapy Results
At 1–3 months: Initial improvements in glucose levels and neuropathy symptoms
At 3–6 months: Measurable HbA1c reduction and C-peptide increase
After 6months: Sustained effects and potential medication reduction under physician supervision.
Reach us directly — WhatsApp and email inquiries are free of charge.
Frequently Asked Questions
Can stem cell therapy cure my diabetes?
We do not use the word “cure.” What clinical evidence demonstrates is that many patients achieve clinical remission — where blood sugar stabilizes to the point where medication can be significantly reduced or paused under physician supervision. The degree of improvement depends on your disease duration, remaining β-cell function, severity of insulin resistance, existing complications, and overall metabolic health.
Is autologous stem cell therapy safe?
Autologous ADMSC therapy has an excellent safety profile. Because we use your own cells, there is no immune rejection risk. A comprehensive 15-year meta-analysis published in Stem Cell Research & Therapy (2021) found no serious adverse events such as death and infection associated with MSC therapy. Japan’s regulatory framework adds an additional layer of safety assurance unmatched globally.
I am worried about complications like blindness or amputation. Can stem cell therapy help?
Yes. One of the most significant benefits of ADMSC therapy is angiogenesis — the formation of new blood vessels. By restoring microcirculation to the eyes, kidneys, nerves, and extremities, stem cell therapy directly opposes the vascular deterioration that leads to these devastating complications. This is something no oral medication can do.
Do you treat both Type 1 and Type 2 Diabetes?
Yes. For T1DM, ADMSCs halt autoimmune destruction and protect remaining β-cells. For T2DM, the focus is on reversing insulin resistance, reducing systemic inflammation, and regenerating β-cell function. Both types benefit substantially from treatment of diabetic complications.
Who is not a candidate for stem cell therapy?
Exclusion criteria are limited but important: active infection, recent or active cancer, severe organ failure, or inability to commit to follow-up monitoring. Most diabetic patients, including those with complications, are suitable candidates. A consultation determines individual eligibility.
Where can I get stem cell treatment for diabetes?
Stem cell treatment for diabetes is available in several countries, but regulatory quality varies significantly. Japan is currently the only country with a dedicated national law governing stem cell therapy — the Act on the Safety of Regenerative Medicine (2014) — which requires government registration, certified cell processing, and ongoing safety monitoring for every treatment. Cell Grand Clinic in Osaka, Japan holds MHLW-certified Regenerative Medicine Provision Plan No. PB5250050 specifically for diabetes and prediabetes. Other countries offering stem cell therapy for diabetes include Mexico, Thailand, Germany, and Panama — but none operate under an equivalent regulatory framework. When evaluating clinics, key questions to ask include: Is the treatment government-registered? Is the cell processing facility certified to GMP standards? Does the physician hold board certification in regenerative medicine?
How much does stem cell therapy for diabetes cost?
The cost of stem cell therapy for diabetes varies by country, clinic, and treatment protocol. In Japan, where treatments are government-regulated and use certified cell processing facilities, pricing reflects the quality control and regulatory compliance involved. At Cell Grand Clinic, treatment costs depend on the number of cells administered, the delivery method (IV drip, local injection, or combination), and whether complication-specific treatments are included. We recommend contacting us directly for a personalized cost estimate based on your medical history and treatment goals — WhatsApp and email inquiries are free of charge.
Can stem cell therapy reverse type 2 diabetes?
Clinical evidence suggests that stem cell therapy can partially reverse the metabolic dysfunction underlying type 2 diabetes — specifically insulin resistance and β-cell exhaustion — though the term “reversal” requires careful qualification. A 2025 meta-analysis found that type 2 diabetes patients were 6.76 times more likely to achieve meaningful glycemic improvement with stem cell therapy compared to controls. The mechanism involves restoration of GLUT4 glucose transporters, repair of insulin signaling pathways (IRS-1/AKT), and suppression of chronic inflammation (NLRP3 inflammasome). Many patients achieve significant HbA1c reduction and medication reduction under physician supervision. However, stem cell therapy is not a guaranteed cure — outcomes depend on disease duration, remaining pancreatic function, and adherence to metabolic management after treatment.
Take Control: From Symptom Management to Regeneration
You have spent years managing your diabetes with medication. The numbers may look acceptable on paper. But if you are honest with yourself, you know the disease is progressing. The complications are advancing. The medication doses are increasing.
There is another path — one that does not replace your medications overnight, but offers something no pill or injection ever could: the opportunity to repair the damage, restore function, and potentially reduce your dependence on lifelong pharmaceutical management.
Stem cell therapy is not about abandoning conventional medicine. It is about adding a regenerative dimension to your treatment — one that works to heal the underlying disease rather than merely suppress its symptoms. This is the future of diabetes care, and it is available today under the strictest regulatory oversight in the world.
At Cell Grand Clinic in Osaka, Japan, we provide personalized stem cell therapy for international patients who are ready to move beyond management — toward regeneration.
📍 Shinsaibashi, Osaka, Japan | ✈️ 50 min from Kansai International Airport (KIX)
🌐 English consultations available | 📋 MHLW Plan No. PB5250050 (Diabetes Mellitus and Prediabetes)
Reach us directly — WhatsApp and email inquiries are free of charge.
References
1. Li H, et al. A meta-analysis on application and prospect of cell therapy in the treatment of diabetes mellitus. Stem Cell Res Ther. 2025;16:249. https://doi.org/10.1186/s13287-025-04377-4
2. Araujo DB, et al. Allogenic adipose tissue-derived stromal/stem cells and vitamin D supplementation in patients with recent-onset type 1 diabetes mellitus: a 3-month follow-up pilot study. Front Immunol. 2020;11:993. https://doi.org/10.3389/fimmu.2020.00993
3. Mikłosz A, Chabowski A. Adipose-derived mesenchymal stem cells therapy as a new treatment option for diabetes mellitus. J Clin Endocrinol Metab. 2023;108(8):1889-1897. https://doi.org/10.1210/clinem/dgad142
4. Elainein H, et al. Therapeutic potential of adipose-derived stem cells for diabetic foot ulcers: a systematic review and meta-analysis. Diabetol Metab Syndr. 2025;17:9. https://doi.org/10.1186/s13098-024-01523-5
5. Du H, et al. Efficacy of stem cell therapy for diabetic kidney disease: a systematic review and meta-analysis. Front Med. 2025;12:1601900. https://doi.org/10.3389/fmed.2025.1601900
6. Wang Y, Yi H, Song Y. The safety of MSC therapy over the past 15 years: a meta-analysis. Stem Cell Res Ther. 2021;12:545. https://doi.org/10.1186/s13287-021-02609-x
Disclaimer: Individual results may vary. This article is for educational purposes and does not constitute medical advice. Stem cell therapy outcomes depend on individual health conditions, disease severity, remaining β-cell function, and other factors. Always consult with a qualified healthcare provider before making treatment decisions. Cell Grand Clinic operates under Japan’s Act on the Safety of Regenerative Medicine with all required government authorizations.
Updated: 2026.05.22



 Inquiries by Email
Inquiries by Email

