- The Molecule That Caught Harvard’s Attention
- What Is NAD+ and Why Does It Decline with Age?
- NAD+ IV Drips — Popular, But Problematic
- NMN — The Smarter Way to Restore NAD+
- Our NMN IV Formulation: N-Pro Med™
- NMN Supplements — Why Quality Varies Dramatically
- IV Drip vs Oral Supplement — When to Use Which
- Dosage, Frequency and What to Expect
- Safety — What 6+ Human Trials Have Confirmed
- Treatment Flow at Cell Grand Clinic
- Common Questions About NMN & NAD+ Therapy
- Ready to Restore Your NAD+ Levels?
- References
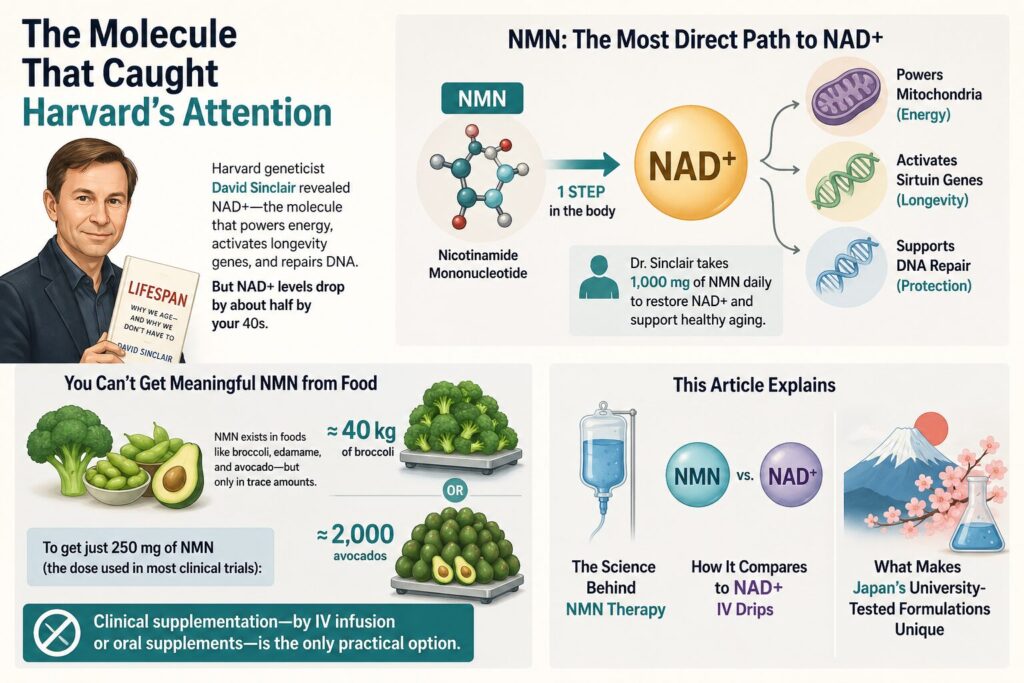
The Molecule That Caught Harvard’s Attention
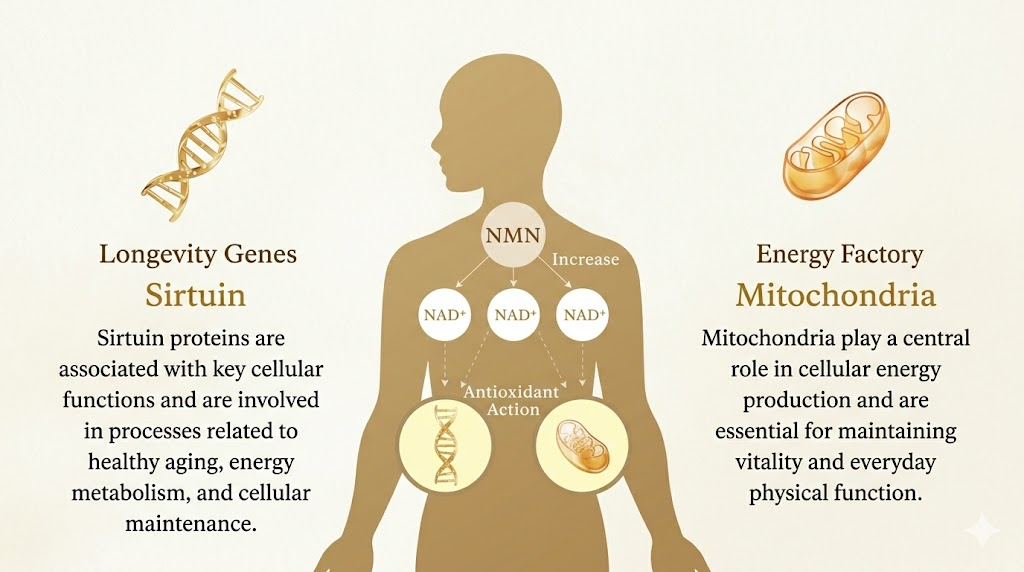
In 2019, Harvard geneticist David Sinclair published Lifespan: Why We Age — and Why We Don’t Have To, a New York Times bestseller translated into 18 languages. The book brought global attention to NAD+ (nicotinamide adenine dinucleotide) — a molecule found in every cell that powers mitochondria, activates sirtuin “longevity genes,” and fuels DNA repair. The problem: NAD+ levels drop by roughly half by your 40s, and with that decline comes reduced energy, slower recovery, and accelerated aging.
NMN (nicotinamide mononucleotide) is the most direct precursor to NAD+. Think of it as the raw material your body converts into NAD+ in a single enzymatic step. Sinclair himself takes 1,000 mg of NMN daily — a fact he has shared publicly in his book and interviews — because replenishing NMN is the most efficient way to refuel sirtuins and restore mitochondrial function at the source.
But you cannot get meaningful NMN from food. It exists naturally in broccoli, edamame, and avocado, but only in trace amounts. To obtain just 250 mg (the dose used in most clinical trials), you would need roughly 40 kg of broccoli or 2,000 avocados in a single day. Clinical supplementation — by IV infusion or oral supplements — is the only practical option.
This article explains the science behind NMN therapy, how it compares to NAD+ IV drips, and what makes Japan’s university-tested formulations different from anything else available today.
What Is NAD+ and Why Does It Decline with Age?
The Role of NAD+ in Energy, DNA Repair and Longevity
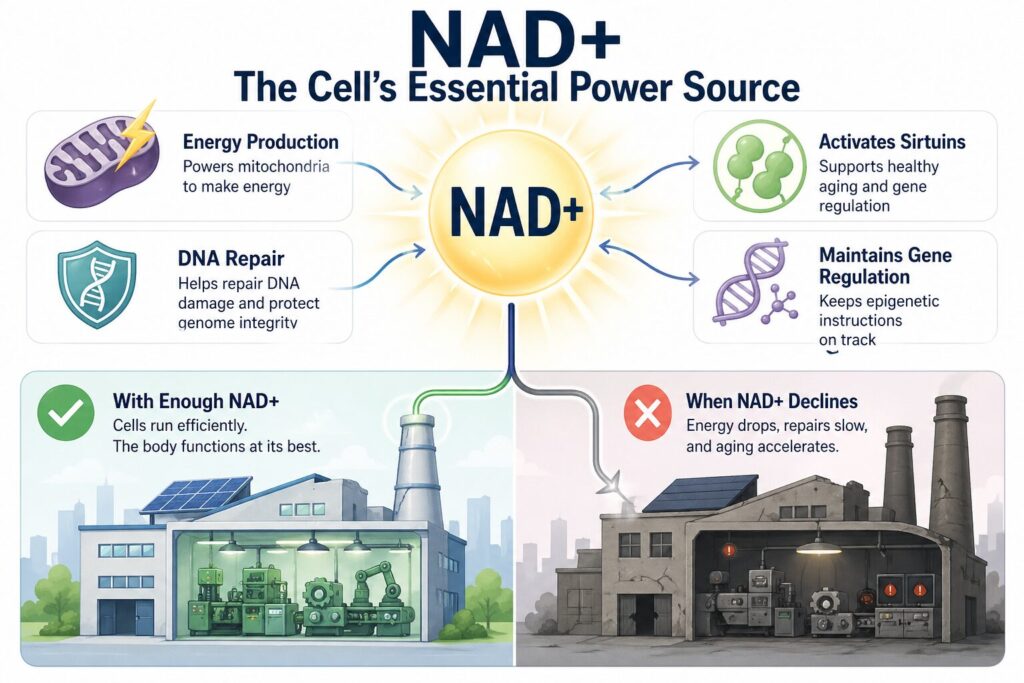
To understand why NMN therapy works, it helps to understand what NAD+ actually does inside your cells. NAD+ serves as an essential helper molecule for more than 500 enzymatic reactions. Beyond powering mitochondrial energy production and activating sirtuins (as described above), NAD+ is also required by PARP enzymes — the cellular emergency repair crew that fixes DNA breaks. Without adequate NAD+, DNA damage accumulates, mitochondria lose efficiency, and the epigenetic instructions that keep genes properly regulated begin to drift.
In simple terms, if your body were a factory, NAD+ would be the electricity. When the power supply drops, every machine in the factory slows down.
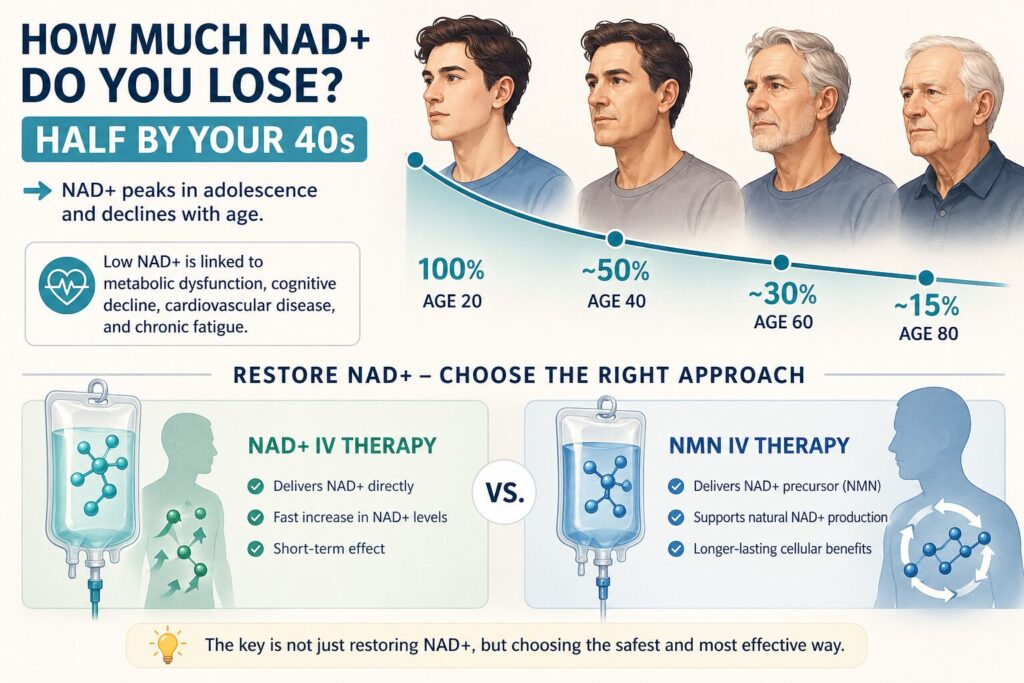
How Much NAD+ Do You Lose? (Half by Your 40s)
NAD+ levels peak during adolescence and decline steadily thereafter. By the age of 40, most people have approximately half the NAD+ they had at 20. By 60, the decline accelerates further. This reduction is strongly correlated with age-related conditions including metabolic dysfunction, cognitive decline, cardiovascular disease, and chronic fatigue.
The question, therefore, is not whether to restore NAD+ but how to do so most effectively and safely. Two clinical approaches exist today: infusing NAD+ directly (NAD+ IV therapy) or infusing its precursor, NMN (NMN IV therapy). The difference between these two approaches is more significant than most people realize.
NAD+ IV Drips — Popular, But Problematic
How NAD+ IV Therapy Works
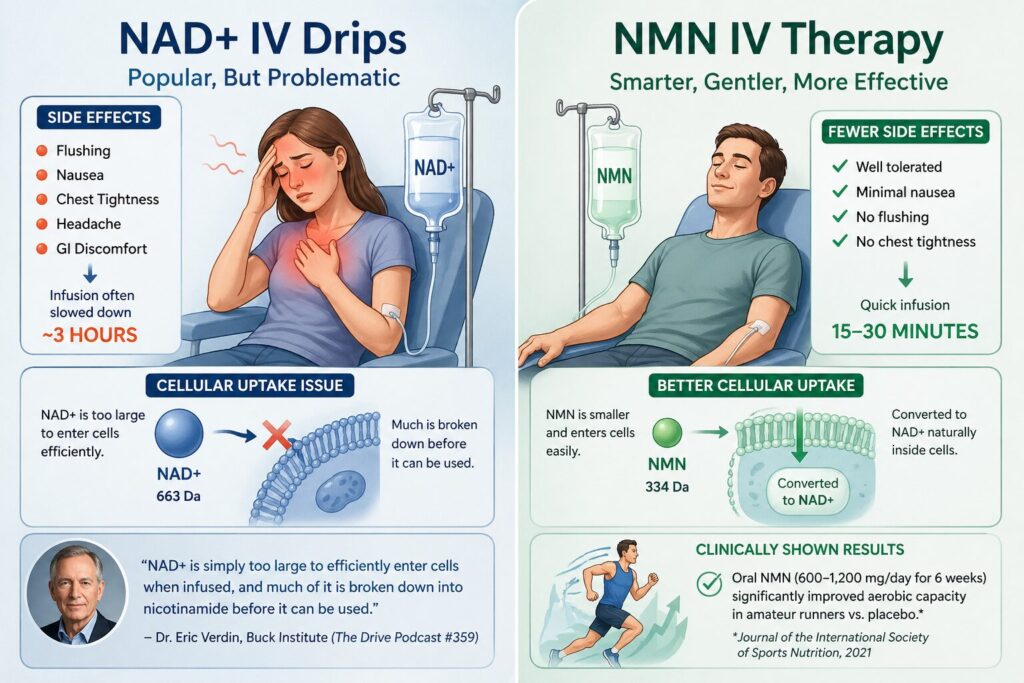
NAD+ IV therapy involves infusing the NAD+ molecule directly into the bloodstream through an intravenous drip. The premise is straightforward: bypass the digestive system and deliver 100% of the dose directly into circulation. It is widely offered at wellness clinics across the United States, Europe, and Southeast Asia, typically as a 2–4 hour infusion session.
The Side-Effect Problem: Flushing, Nausea and Chest Tightness
Despite its popularity, NAD+ IV therapy is associated with a range of uncomfortable side effects. A systematic review of clinical evidence on NAD+ interventions found that the most commonly reported adverse effects include skin flushing (from rapid prostaglandin release), nausea, gastrointestinal discomfort, headaches, and chest tightness. These effects frequently require the infusion rate to be slowed, extending treatment duration to 3 hours per session.
In contrast, NMN IV therapy is typically completed in 15–30 minutes and produces significantly fewer adverse effects, because NMN is converted to NAD+ gradually through natural enzymatic pathways rather than flooding the system with a large molecule all at once.
The Cellular Uptake Problem — “NAD+ Is Too Large to Enter Cells”
Perhaps the more fundamental concern is whether intravenous NAD+ actually reaches the cells where it is needed. NAD+ has a molecular weight of 663 Da — substantially larger than NMN (334 Da). While NAD+ can elevate plasma levels when injected, its ability to cross cell membranes and enter the intracellular space is limited.
Dr. Eric Verdin, President of the Buck Institute for Research on Aging — one of the world’s leading longevity research institutions — has publicly expressed skepticism about intravenous NAD+ therapy. In a conversation with physician Peter Attia on The Drive podcast (episode #359), Dr. Verdin explained that NAD+ is simply too large a molecule to efficiently enter cells when infused, and that much of it is broken down into nicotinamide before it can be used. He recommended oral precursors like NMN or NR as a more scientifically sound approach for most people.
Furthermore, a 2021 randomized trial published in the Journal of the International Society of Sports Nutrition showed that oral NMN supplementation (600 mg and 1,200 mg/day for 6 weeks) significantly increased aerobic capacity in amateur runners compared to placebo. This is notable because it demonstrates that NMN’s NAD+-boosting effect translates into measurable physiological improvements — something NAD+ IV therapy has not been shown to achieve in a controlled trial setting.
NMN — The Smarter Way to Restore NAD+
What Is NMN?
Nicotinamide mononucleotide (NMN) is a naturally occurring molecule and a direct precursor to NAD+. As discussed above, it exists in trace amounts in common foods, but not in quantities anywhere close to what clinical studies use. This is why supplementation is necessary.
Inside the body, NMN is converted to NAD+ through a single enzymatic step catalyzed by NMNAT enzymes. This conversion happens efficiently within cells, meaning NMN effectively “refills” NAD+ from the inside. NMN enters cells via the Slc12a8 transporter, a dedicated cellular gateway identified in 2019, which explains why NMN supplementation consistently raises intracellular NAD+ levels in both animal and human studies.
NMN vs NAD+ IV: Head-to-Head Comparison
| Feature | NMN IV Therapy | NAD+ IV Therapy |
| Molecular Weight | 334 Da (smaller, enters cells) | 663 Da (larger, limited cell entry) |
| Cell Entry Mechanism | Slc12a8 transporter (dedicated) | Limited; mostly broken down extracellularly |
| Conversion to NAD+ | Gradual, via natural NMNAT pathway | Direct, but much is degraded to nicotinamide |
| Infusion Duration | 15–30 minutes | 2–4 hours |
| Common Side Effects | Minimal (mild warmth in some cases) | Flushing, nausea, chest tightness, headache |
| Human Clinical Trials | 6+ published RCTs (oral + IV) | Very limited (mostly addiction treatment) |
| University IV Safety Data | Yes (Hyogo Medical University, Japan) | No university-validated IV formulation |
Reach us directly — WhatsApp and email inquiries are free of charge.
What the Clinical Trials Say About NMN
Unlike NAD+ IV therapy, which has very limited controlled clinical evidence, NMN has been studied in multiple randomized, placebo-controlled trials:
Keio University / Washington University (Irie et al., 2020): The first human clinical study of oral NMN. Ten healthy Japanese men aged 40–60 received single doses of 100, 250, or 500 mg NMN. No clinically significant adverse effects were observed. NMN metabolites increased dose-dependently in the blood, confirming safe absorption and metabolism.
Okabe et al. (2022), Frontiers in Nutrition — RCT: Thirty healthy adults took 250 mg/day of oral NMN or placebo for 12 weeks. Blood NAD+ levels rose significantly in the NMN group compared to placebo, with no adverse events reported. This confirmed that oral NMN safely and efficiently increases circulating NAD+.
Katayoshi et al. (2023), Scientific Reports — RCT: Thirty-six healthy middle-aged adults received 250 mg/day of NMN or placebo for 12 weeks. NAD+ metabolites increased significantly, and pulse wave velocity (an indicator of arterial stiffness) showed a favorable declining trend in the NMN group. This suggests NMN may benefit cardiovascular health beyond simple NAD+ replenishment.
Yoshino et al. (2021), Science: Twenty-five postmenopausal women with prediabetes received 250 mg/day NMN or placebo for 10 weeks. The NMN group showed significantly improved skeletal muscle insulin sensitivity, with enhanced Akt and mTOR signaling confirmed by muscle biopsy. This landmark study in one of the world’s most prestigious journals provided the first clinical evidence that NMN improves metabolic function in humans.
Hyogo Medical University IV Safety Study: A clinical study led by Professor Akinobu Goto confirmed the safety of 300 mg intravenous NMN administration. The IV formulation did not affect electrocardiogram readings, pulse rate, blood pressure, or metabolic markers in the liver, heart, pancreas, or kidney. Blood NAD+ levels increased by approximately 20%, and blood triglycerides were significantly reduced.
Across all published human trials, no serious adverse events have been reported with NMN at doses up to 1,200 mg/day (oral) or 300 mg (IV).
Our NMN IV Formulation: N-Pro Med™
The Only NMN IV Formulation Tested in a Japanese University Clinical Study
Cell Grand Clinic uses N-Pro Med™, manufactured by Nordeste Inc. This is currently the only NMN IV formulation whose safety and efficacy have been validated through a clinical study at a Japanese medical university (Hyogo Medical University, led by Professor Akinobu Goto).
The key findings from this study were:
• Blood NAD+ levels increased by approximately 20% following a single IV infusion
• Blood triglyceride levels were significantly reduced — suggesting potential benefits for fatty liver disease and metabolic syndrome
• No effects on ECG, pulse rate, blood pressure, or organ function markers
Why the Formulation Matters
NMN IV therapy is not yet an approved pharmaceutical treatment in any country. In Japan, physicians are legally permitted to administer unapproved formulations under their own clinical judgment. This means the quality, purity, and safety profile of the NMN formulation depends entirely on the manufacturer and the clinic’s selection process.
Many clinics in Japan and abroad use NMN IV formulations that have never been tested in any human clinical study. Cell Grand Clinic specifically chose N-Pro Med™ because it is the only formulation backed by published university-level safety data. For a molecule being infused directly into your bloodstream, we believe this distinction matters.
NMN Supplements — Why Quality Varies Dramatically
Chemical Synthesis vs Fermentation: The α-NMN Contamination Risk
NMN raw materials are produced by two fundamentally different methods: chemical synthesis and enzymatic fermentation. Understanding this difference is critical because the method determines the molecular composition of what you are consuming.
Chemically synthesized NMN is cheaper to mass-produce, but carries two significant risks. First, unless specifically labeled as β-NMN, the product may contain α-NMN — a structural variant that has not been studied for safety or efficacy in humans. Second, chemical synthesis can produce D-form NMN (an optical isomer) rather than the naturally occurring L-form. The human body uses only L-form β-NMN, which is the same form produced by fermentation.
Fermentation-derived NMN produces exclusively L-form β-NMN, the bioidentical molecule found naturally in the body. The disadvantage is cost — fermentation-grade NMN is significantly more expensive. However, for a supplement you take daily for long-term health, the difference in raw material quality is substantial.
Investigative reports in both the United States and Japan have revealed that some commercial NMN supplements contain no detectable NMN at all. In Japan, an analysis by a major newspaper found that two out of eight popular NMN products tested contained zero measurable NMN. This underscores the importance of choosing products with verified purity from reputable sources.
Cell Grand Clinic’s NMN supplement uses exclusively fermentation-derived L-form β-NMN with a verified purity of 99.91% by absolute quantitative analysis.
Our nanoPDS® Technology: 3–4× Higher Absorption
Even with high-purity NMN, conventional oral supplements face a fundamental challenge: the first-pass effect. After swallowing, the active ingredient must survive stomach acid, pass through the intestinal wall, and navigate liver metabolism before reaching systemic circulation. At each step, a portion of the NMN is degraded or deactivated.
To address this, Cell Grand Clinic developed an original NMN supplement using nanoPDS® (Particle Delivery System) technology — a patented nano-encapsulation method developed in collaboration with a Japanese national university through the Next-Generation Absorption Research Association. The technology compresses NMN into nano-sized particles coated with an oil layer that protects against gastric acid degradation, enabling efficient absorption through the intestinal wall.
Independent testing has shown that nanoPDS® delivers 3–4 times higher absorption compared to conventional NMN capsules, significantly increasing the amount of NMN that reaches the bloodstream.
Reach us directly — WhatsApp and email inquiries are free of charge.
IV Drip vs Oral Supplement — When to Use Which
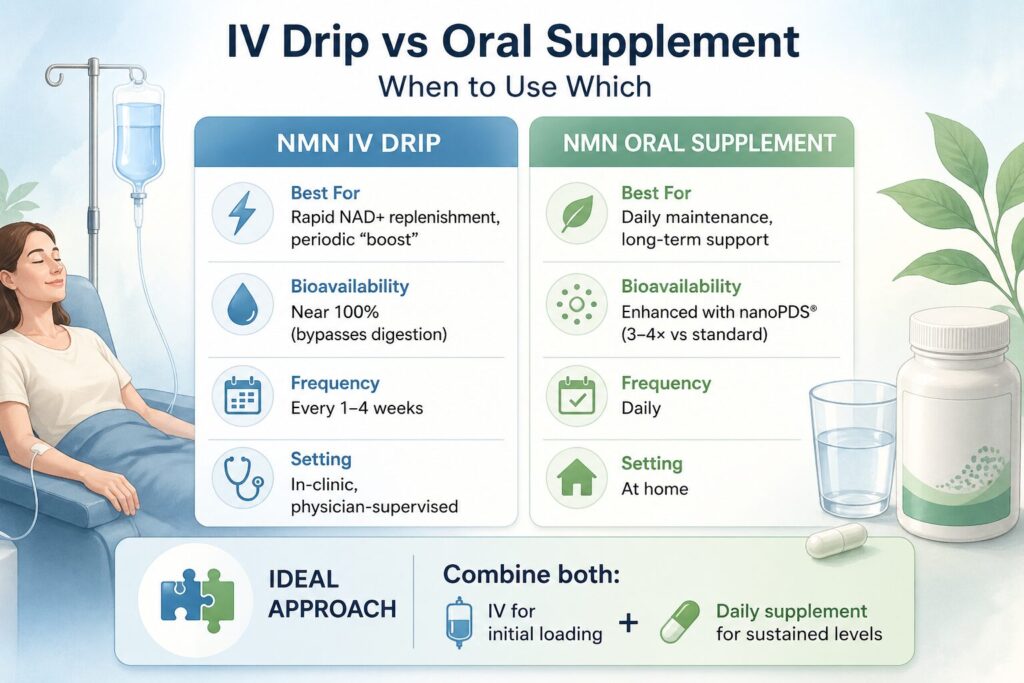
NMN IV therapy and NMN oral supplementation are not competing treatments — they serve complementary roles:
| NMN IV Drip | NMN Oral Supplement | |
| Best For | Rapid NAD+ replenishment, periodic “boost” | Daily maintenance, long-term support |
| Bioavailability | Near 100% (bypasses digestion) | Enhanced with nanoPDS® (3–4× vs standard) |
| Frequency | Every 1–4 weeks | Daily |
| Setting | In-clinic, physician-supervised | At home |
| Ideal Approach | Combine both: IV for initial loading + daily supplement for sustained levels |
Dosage, Frequency and What to Expect
NMN IV Drip Protocol
At Cell Grand Clinic, NMN IV therapy is administered as a 300 mg infusion over approximately 15–30 minutes. For initial treatment, a series of sessions at 1–2 week intervals is typically recommended, followed by maintenance sessions every 2–4 weeks depending on individual goals and baseline NAD+ status.
NMN IV therapy is a same-day treatment — no overnight stay, no recovery period, and no restrictions on normal activities afterward. Many patients combine NMN IV with other treatments available at the clinic, such as stem cell therapy or exosome therapy, as part of a comprehensive longevity protocol.
NMN Supplement Dosage
Clinical trials have established the safety of oral NMN at doses ranging from 250 mg to 1,200 mg per day, with the most commonly studied dose being 250 mg/day. Higher doses (up to 2,000 mg/day) have been tested without serious adverse events in Harvard Medical School-affiliated studies.
For most adults seeking anti-aging and metabolic benefits, 250–500 mg/day of high-quality NMN is a reasonable starting point. Our nanoPDS® supplement is designed for once-daily dosing.
Combining IV + Supplement for Optimal Results
The most effective NMN strategy is a two-phase approach: begin with a series of IV infusions to rapidly restore NAD+ levels (the “loading phase”), then maintain those levels with a daily oral supplement (the “maintenance phase”). This mirrors the approach used in other areas of medicine where an initial intensive treatment is followed by ongoing maintenance therapy.
Safety — What 6+ Human Trials Have Confirmed
No Serious Adverse Events Across All Published Trials
Across all completed human clinical trials of NMN — including studies conducted at Keio University (Japan), Washington University (USA), Hyogo Medical University (Japan), Hiroshima University (Japan), and multiple sites in China — no serious adverse events have been reported. Mild, transient effects such as slight stomach discomfort (with oral supplementation on an empty stomach) have been noted in a small number of participants, but these resolve without intervention.
A comprehensive review of NMN safety data published in 2023 concluded that NMN supplementation appears to be well tolerated in healthy human subjects at doses tested to date, and that the safety profile supports continued clinical investigation.
Treatment Flow at Cell Grand Clinic
Step 1 — Consultation: A physician reviews your health history, current concerns, and treatment goals. Blood work may be recommended to establish baseline markers.
Step 2 — Treatment Plan: Based on your consultation, the physician recommends an NMN protocol (IV only, supplement only, or the combined loading + maintenance approach). If you are combining NMN with other treatments such as stem cell therapy, the physician will design an integrated protocol.
Step 3 — NMN IV Infusion: The N-Pro Med™ formulation is administered via IV drip over approximately 15–30 minutes in a comfortable private treatment room. No anesthesia or special preparation is required.
Step 4 — Supplement Prescription: If included in your plan, the physician prescribes our nanoPDS® NMN supplement for daily maintenance between IV sessions.
Step 5 — Follow-Up: Periodic blood testing to monitor NAD+ levels and other biomarkers allows us to adjust your protocol over time for optimal results.
NMN therapy pairs powerfully with other regenerative treatments such as stem cell thrapy. Explore our detailed guides:
Reverse Aging: How Stem Cell IV Therapy Actually Works Learn How Skin Rejuvenation Beyond Fillers: Rebuild Collagen From Within Learn More AGA & FAGA: A Regenerative Approach to Hair Loss Learn More Drug-Free ED Treatment: Stem Cells Without Pills or Implants Learn More Stem Cell Therapy for Diabetes: A Regenerative Approach to Glycemic Control Learn More Improving Atherosclerosis: Stem Cell Therapy for Vascular & Cardiac Health Learn MoreCommon Questions About NMN & NAD+ Therapy
What is NMN and how does it restore NAD+ levels?
NMN (nicotinamide mononucleotide) is a naturally occurring molecule and the most direct precursor to NAD+, a coenzyme essential for energy production, DNA repair, and sirtuin activation in every cell. NMN is converted to NAD+ through a single enzymatic step via NMNAT enzymes and enters cells through the dedicated Slc12a8 transporter. By age 40, NAD+ levels drop to approximately half of what they were at age 20, making supplementation through IV infusion or oral supplements the only practical way to restore them.
Why is NMN IV therapy considered better than NAD+ IV drips?
NMN has a molecular weight of 334 Da — roughly half that of NAD+ (663 Da) — allowing it to enter cells far more efficiently via a dedicated transporter. NAD+ IV drips frequently cause flushing, nausea, chest tightness, and require 2–4 hours per session, whereas NMN IV therapy is typically completed in 15–30 minutes with minimal side effects. Dr. Eric Verdin, President of the Buck Institute for Research on Aging, has publicly stated that NAD+ is too large to efficiently enter cells when infused. NMN also has stronger clinical evidence, with 6+ published human trials compared to very limited controlled data for NAD+ IV.
How long does an NMN IV drip session take?
An NMN IV drip at Cell Grand Clinic takes approximately 15–30 minutes per session using the N-Pro Med formulation at a dose of 300 mg. This is significantly shorter than NAD+ IV therapy, which typically requires 2–4 hours. No anesthesia, overnight stay, or recovery period is needed — patients can resume normal activities immediately after treatment.
Is NMN therapy safe? What do clinical trials show?
Across all published human clinical trials — including studies at Keio University, Washington University, Hyogo Medical University, and Hiroshima University — no serious adverse events have been reported with NMN at doses up to 1,200 mg/day orally or 300 mg intravenously. The N-Pro Med IV formulation used at Cell Grand Clinic is the only NMN IV product whose safety has been validated through a clinical study at a Japanese medical university (Hyogo Medical University), confirming no effects on ECG, blood pressure, or organ function.
What is the difference between NMN IV drip and NMN supplements?
NMN IV therapy delivers the molecule directly into the bloodstream with near-100% bioavailability, providing a rapid NAD+ boost in a single 15–30 minute session. Oral NMN supplements are taken daily for sustained maintenance but face absorption challenges from stomach acid and first-pass liver metabolism. The most effective strategy combines both: an initial series of IV infusions to rapidly restore NAD+ levels (the loading phase), followed by a daily oral supplement for ongoing maintenance.
Can NMN therapy help with anti-aging and longevity?
Clinical evidence supports NMN’s role in addressing age-related decline. A 2021 study in Science showed NMN improved skeletal muscle insulin sensitivity in prediabetic women. Separate trials demonstrated increased aerobic capacity in runners and improved arterial stiffness markers. NMN works by replenishing NAD+, which activates sirtuin longevity genes, fuels mitochondrial energy production, and supports DNA repair — the fundamental biological processes that deteriorate with aging.
How does Cell Grand Clinic’s NMN supplement achieve higher absorption?
Cell Grand Clinic developed an NMN supplement using nanoPDS (Particle Delivery System) technology — a patented nano-encapsulation method created in collaboration with a Japanese national university. This technology compresses NMN into nano-sized particles coated with a protective oil layer that resists gastric acid degradation. Independent testing shows nanoPDS delivers 3–4 times higher absorption compared to conventional NMN capsules. The raw material is exclusively fermentation-derived L-form beta-NMN with 99.91% verified purity.
Ready to Restore Your NAD+ Levels?
Cell Grand Clinic is located in Shinsaibashi, Osaka — one of Japan’s most accessible medical destinations. Our NIH-trained Medical Director, Dr. Yuichi Wakabayashi, personally oversees every NMN treatment protocol.
Reach us directly — WhatsApp and email inquiries are free of charge.
References
1. Irie J, et al. Effect of oral administration of nicotinamide mononucleotide on clinical parameters and nicotinamide metabolite levels in healthy Japanese men. Endocr J. 2020;67(2):153-160. https://doi.org/10.1507/endocrj.EJ19-0313
2. Okabe K, et al. Oral Administration of Nicotinamide Mononucleotide Is Safe and Efficiently Increases Blood Nicotinamide Adenine Dinucleotide Levels in Healthy Subjects. Front Nutr. 2022;9:868640. https://doi.org/10.3389/fnut.2022.868640
3. Katayoshi T, et al. Nicotinamide adenine dinucleotide metabolism and arterial stiffness after long-term nicotinamide mononucleotide supplementation: a randomized, double-blind, placebo-controlled trial. Sci Rep. 2023;13:2786. https://doi.org/10.1038/s41598-023-29787-3
4. Yoshino M, et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021;372(6547):1224-1229. https://doi.org/10.1126/science.abe9985
5. Goto A, et al. Safety and metabolic effects of intravenous NMN administration (N-Pro Med™). Hyogo Medical University [clinical study data; referenced via Nordeste Inc. product documentation].
6. Mills KF, et al. Long-Term Administration of Nicotinamide Mononucleotide Mitigates Age-Associated Physiological Decline in Mice. Cell Metab. 2016;24(6):795-806. https://doi.org/10.1016/j.cmet.2016.09.013
7. Imai S. The first human clinical study for NMN has started in Japan. npj Aging Mech Dis. 2017;3:14. https://doi.org/10.1038/s41514-017-0015-7
8. Liao B, et al. Nicotinamide mononucleotide supplementation enhances aerobic capacity in amateur runners: a randomized, double-blind study. J Int Soc Sports Nutr. 2021;18(1):54. https://doi.org/10.1186/s12970-021-00442-4
9. Yi L, et al. The efficacy and safety of β-nicotinamide mononucleotide (NMN) supplementation in healthy middle-aged adults: a randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial. GeroScience. 2023;45(1):29-43. https://doi.org/10.1007/s11357-022-00705-1
10. Nadeeshani H, et al. Nicotinamide mononucleotide (NMN) as an anti-aging health product – Promises and safety concerns. J Adv Res. 2022;37:142-150. https://doi.org/10.1016/j.jare.2021.08.003
Updated: 2026.05.07



 Inquiries by Email
Inquiries by Email

