- Opening: Regeneration Without the Wait
- What Is Exosome Therapy?
- How Exosome Therapy Works — The Science Behind the Treatment
- Clinical Evidence — What the Research Shows
- Available Exosome Treatment Options
- Exosome Therapy vs. Stem Cell Therapy — Understanding the Difference
- Who Is a Good Candidate for Exosome Therapy?
- Why Japan — and Why Cell Grand Clinic?
- Frequently Asked Questions About Exosome Therapy
- The Next Step: Regenerate While You’re Here
- References
Opening: Regeneration Without the Wait
You’re in Japan for two weeks. You want visible skin improvement — tighter, brighter, refreshed. Or maybe hair density. Or joint relief. But a multi-week stem cell culture program doesn’t fit your timeline.
What if regeneration could happen today?
That’s exosome therapy: the express route to cellular renewal. Same day. No culture period. No waiting. Just mesenchymal stem cell-derived exosomes — tiny biological messengers — injected directly into your skin, scalp, joints, or delivered intravenously for whole-body benefit. You walk in. You walk out with regeneration already underway.
What Is Exosome Therapy?
Exosome therapy is a regenerative treatment that uses nano-sized vesicles (30–150 nm) derived from mesenchymal stem cells to deliver growth factors, cytokines, and microRNA directly to damaged tissue — promoting repair, reducing inflammation, and stimulating cellular renewal without cell transplantation.
But what are exosomes, really?
Imagine stem cells as a factory. They produce and release tiny cargo containers called exosomes — small enough at 30–150 nanometers that thousands fit on a single human hair. Inside each one: growth factors, microRNA, anti-inflammatory proteins, and signals that tell your cells to repair and regenerate.
Unlike stem cell therapy, where living cells are transplanted, exosome therapy delivers only the instructions — the molecular messengers. Your own cells read these instructions and get to work. No integration risk. No cell rejection. Just pure paracrine signaling: one cell’s way of telling another, “Heal yourself.”
This distinction is why exosomes are the same-day option. There’s no cell culture needed. The exosomes already exist in a high-concentration, high-purity form. Draw them up. Deliver them. Start the regeneration.
How Exosome Therapy Works — The Science Behind the Treatment
Exosome therapy operates through paracrine signaling — the biological mechanism by which stem cells communicate repair instructions to surrounding tissue via extracellular vesicles, triggering regeneration without requiring cell transplantation.
Mesenchymal stem cell-derived exosomes carry three main classes of cargo:
Growth factors are proteins that stimulate cell division, differentiation, and tissue repair. MicroRNA are genetic regulators that fine-tune cell behavior and collagen production. Anti-inflammatory cytokines are proteins that reduce swelling, calm immune overreaction, and create an environment where healing happens faster.
When exosomes reach target tissue — whether skin, hair follicles, cartilage, or blood vessels — they dock with receptors on damaged or aging cells. The cargo is released. That cell “reads” the message and responds: produce more collagen, reduce inflammation, multiply, differentiate, vascularize.
A 2026 systematic review by Wang and colleagues in Molecular and Cellular Probes, examining exosome mechanisms across multiple studies, confirmed four key pathways. Exosomes facilitate wound healing through immunomodulation — reducing pro-inflammatory cytokines (IL-6, TNF-α) while promoting anti-inflammatory signaling — in simpler terms, they calm down the molecules that cause redness and swelling, allowing your body to focus on repair instead of fighting itself. They stimulate cellular differentiation, signaling fibroblasts to increase collagen I and III production — meaning they tell skin-building cells to make more of the protein that keeps skin firm and elastic. They promote angiogenesis — triggering endothelial cells to form new blood vessels — which means better blood flow and oxygen delivery to healing tissue. And they modulate collagen remodeling, upregulating matrix metalloproteinase inhibitors while promoting new collagen synthesis — essentially preventing old collagen from breaking down too fast while building new, organized collagen, resulting in youthful skin architecture.
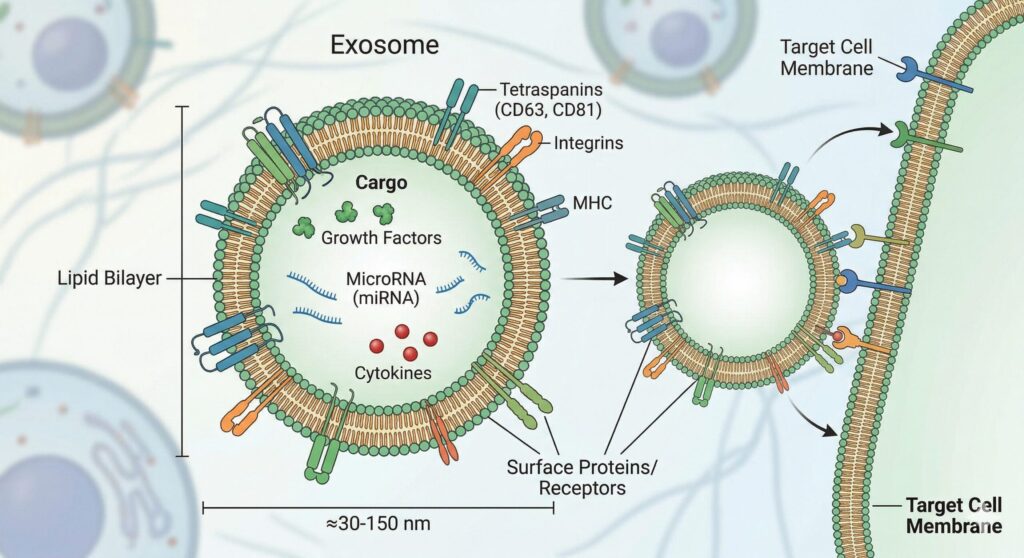
The following diagram illustrates how exosome therapy works from preparation to regeneration at the cellular level.
Clinical Evidence — What the Research Shows
Exosome therapy is supported by a growing body of human clinical trial data demonstrating efficacy and safety across skin rejuvenation, hair regeneration, and systemic applications — with systematic reviews now confirming consistent outcomes.
Skin Rejuvenation
A 2026 systematic review by Flores Rodríguez and colleagues, published in Cureus, analyzed 19 human clinical studies of exosome-based therapies for skin rejuvenation — the largest PRISMA-compliant review (meaning it followed the gold-standard methodology for analyzing multiple studies together) of its kind to date. Across these studies, exosome treatments demonstrated consistent improvements in wrinkle depth, skin elasticity, hydration, pigmentation, and overall texture. Delivery methods ranged from intradermal injection and microneedling to topical application, with mesenchymal stem cell-derived exosomes showing the strongest results. Individual studies reported wrinkle reductions of approximately 15–20%, pore area decreases of up to 17.9%, and measurable gains in skin hydration and barrier function — meaning patients experienced visibly smoother, tighter, and more radiant skin within weeks of treatment. The review concluded that exosome-based therapies show “encouraging early clinical effects” for skin rejuvenation.
Hair Regeneration
A 2025 systematic review by Al Ameer and colleagues in Clinical, Cosmetic and Investigational Dermatology examined 11 clinical studies — including two randomized controlled trials — evaluating exosomes for hair regeneration. All 11 studies demonstrated improvements in at least one hair parameter. MSC-derived exosomes produced hair density gains of 9.5 to 35 hairs/cm² and hair thickness increases of up to 13.01 µm — meaning a patient with thinning hair across the crown could expect noticeably fuller, thicker hair in the treated areas within 12 weeks. No serious adverse events were reported across any of the reviewed studies.
Safety Profile
A 2024 systematic review and meta-analysis by van Delen and colleagues, examining 21 clinical reports of exosome-based therapy, found a serious adverse event rate of only 0.7% — meaning that out of every 1,000 patients treated, only about 7 experienced a serious side effect. The overall adverse event rate was 4.4%, predominantly mild and self-resolving: transient injection site erythema (temporary redness at the injection spot), minimal swelling, mild fatigue — the kind of effects that go away on their own within a few days. No significant difference in serious adverse events were observed between autologous and allogeneic exosome preparations — in plain terms, exosomes sourced from carefully screened donors are just as safe as those derived from your own cells.
Non-Invasive Delivery Routes
A 2026 review by Ayanji and colleagues in the Journal of Controlled Release examined emerging non-invasive exosome delivery methods. The study highlighted intranasal (nasal spray) administration as particularly promising for CNS and systemic applications, as this route bypasses hepatic metabolism and delivers exosomes directly to the brain via olfactory and trigeminal nerve pathways — opening the door to at-home exosome protocols for cognitive and systemic support.
The following table summarizes the key studies that form the clinical evidence base for exosome therapy. Flores Rodríguez et al. (2026) reviews 19 skin rejuvenation studies showing consistent improvements across multiple skin parameters. Al Ameer et al. (2025) covers 11 hair regeneration studies with meaningful density and thickness gains. Van Delen et al. (2024) establishes safety with only a 0.7% serious adverse event rate.
| Study | Year | Design | Studies (n) | Key Finding |
|---|---|---|---|---|
| Flores Rodríguez et al. Cureus | 2026 | Systematic review (PRISMA) | 19 | Consistent wrinkle, elasticity, hydration, and pigmentation improvements; wrinkle reduction 15–20% |
| Al Ameer et al. Clin Cosmet Investig Dermatol | 2025 | Systematic review (incl. 2 RCTs) | 11 | Hair density +9.5–35 hairs/cm²; thickness up to +13.01 µm; no serious AEs |
| Van Delen et al. J Extracell Vesicles | 2024 | Systematic review & meta-analysis | 21 | 0.7% serious adverse events; 4.4% any adverse event; no safety difference autologous vs allogeneic |
| Wang et al. Mol Cell Probes | 2026 | Systematic review | 20+ | Exosomes enhance wound healing via immunomodulation, collagen remodeling, and angiogenesis |
| Ayanji et al. J Control Release | 2026 | Review | — | Intranasal delivery optimal for CNS application; bypasses hepatic metabolism |
All studies used human subjects. Full references with DOI links are provided at the end of this article.
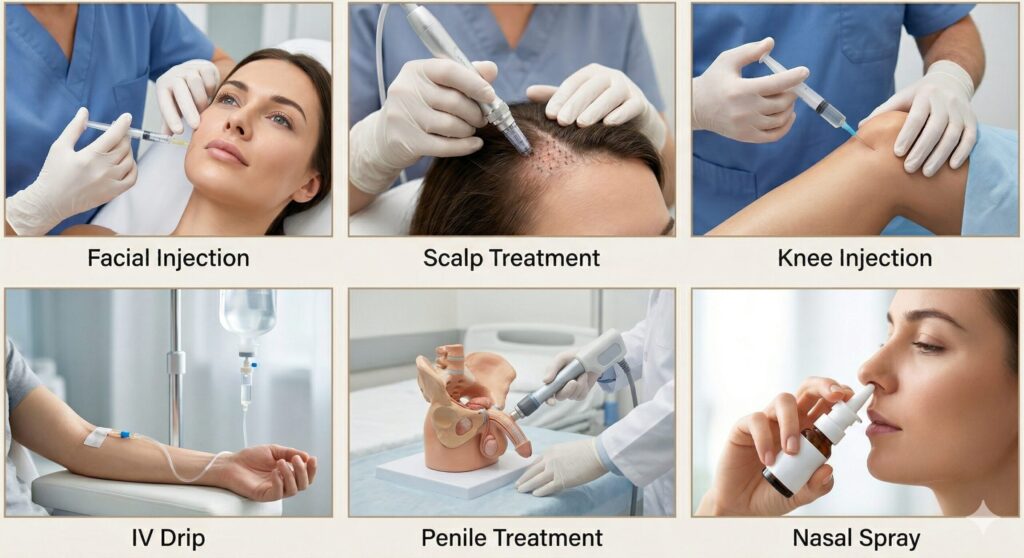
Available Exosome Treatment Options
Exosome therapy encompasses multiple delivery methods — each targeting specific tissues or providing systemic benefit — all available same-day without cell culture, making it an adaptable treatment platform for aesthetic, orthopedic, and whole-body regenerative goals.
IV Drip (Systemic)
IV exosome therapy delivers exosomes intravenously for whole-body circulation and systemic anti-aging benefits. When exosomes enter the bloodstream, they circulate throughout your body. Tissues that need repair — aging skin, worn cartilage, fatigued organs — absorb and process these exosomes. The result: systemic regeneration from a single session.
Use cases include general anti-aging, overall wellness enhancement, joint health, cognitive support, and energy and vitality. Typical protocol: 100–200 million exosomes in a 50 mL IV drip, 20–30 minute infusion.
Facial Injection
Exosomes injected directly into facial skin stimulate collagen production, reduce wrinkles, improve texture, and brighten tone — all within one appointment. Over 2–4 weeks, fibroblasts ramp up collagen production. Inflammation subsides. Skin becomes visibly tighter, smoother, more radiant. The 2026 Flores Rodríguez systematic review of 19 studies confirmed these outcomes across multiple delivery methods.
Use cases include fine lines and wrinkles, loss of firmness, uneven tone, post-acne scarring, and general skin rejuvenation. Typical protocol: 50–100 million exosomes per treatment; single session or 2–3 sessions spaced 2 weeks apart.
Scalp Injection
Exosomes injected into the scalp stimulate dormant hair follicles, increase blood flow, and promote thicker, denser hair growth. Hair loss and thinning are rooted in inflammation and reduced nutrient delivery to follicles. Exosomes address both — reducing scalp inflammation and stimulating growth factor signaling in follicle stem cells. The Al Ameer systematic review of 11 studies showed density gains of 9.5–35 hairs/cm² within 12 weeks.
Use cases include androgenetic alopecia (male/female pattern baldness), early thinning, post-stress hair loss, and hair maintenance. Typical protocol: 50–100 million exosomes distributed across the scalp via fine-needle injection; 2–4 sessions every 2–4 weeks.
Knee Injection
Exosomes injected into the knee joint reduce inflammation, provide cartilage support, and improve mobility for mild-to-moderate joint discomfort. Osteoarthritis is driven by cartilage loss and chronic inflammation. Exosomes can’t reverse severe degeneration, but they can reduce pain, stabilize function, and slow progression by signaling chondrocytes to produce more protective matrix.
Use cases include mild-to-moderate osteoarthritis, post-injury joint pain, athletic joint maintenance, and cartilage protection. Typical protocol: 50–100 million exosomes delivered intra-articularly under ultrasound guidance; often 1–2 sessions. For more advanced joint conditions, stem cell therapy for knee osteoarthritis may offer deeper regeneration.
Penile Injection
Exosomes injected into penile tissue promote endothelial function, angiogenesis, and smooth muscle response — supporting erectile function and tissue regeneration. Erectile dysfunction often involves reduced blood flow and endothelial dysfunction. Exosomes stimulate VEGF (vascular endothelial growth factor) and other pro-angiogenic signals, improving vascular function and tissue elasticity.
Use cases include mild-to-moderate erectile dysfunction, post-prostate surgery ED, and tissue regeneration for sexual function. Typical protocol: 20–50 million exosomes injected intracavernously; 1–2 sessions. Patients with more advanced ED may benefit from stem cell therapy for erectile dysfunction, which provides cell integration for longer-lasting results.
Nasal Spray (At-Home)
Exosome nasal spray is a take-home option for continued regenerative benefit between clinical treatments — ideal for CNS, cognitive, and systemic support. Intranasal delivery bypasses the blood-brain barrier, allowing exosomes to reach the central nervous system directly. This route is supported by recent research from Ayanji and colleagues (2026) in the Journal of Controlled Release, which identified intranasal administration as particularly promising for non-invasive CNS delivery.
Use cases include cognitive support, ongoing systemic wellness, convenient at-home maintenance, and post-treatment extension. Typical protocol: 2–3 sprays per nostril daily.
The Critical Differentiator: Quality and Concentration
Across all applications, one factor determines success: exosome quality and concentration. High-quality means verified exosome concentration (typically 100–200 million per mL for clinical-grade preparations), rigorous characterization through size and surface marker profiling, single-donor sourcing for consistency and traceability, and sterility testing at every production lot. Low-concentration or poorly characterized exosomes may show minimal benefit. High-concentration, CPC-grade exosomes show robust clinical outcomes.
Exosome Therapy vs. Stem Cell Therapy — Understanding the Difference
Exosome therapy and stem cell therapy are related but distinct regenerative tools — exosomes deliver molecular repair signals for same-day, lower-cost improvement, while stem cell therapy provides deeper, longer-lasting regeneration through cell integration for severe or chronic conditions.
The core distinction is speed versus depth. Exosome therapy is available same-day with no culture period, making it ideal for mild conditions, aesthetic enhancement, and maintenance. Stem cell therapy requires approximately 7 weeks of culture and delivers deeper regeneration through cell integration for severe or chronic conditions. Many patients combine both — stem cells as primary treatment and exosomes for ongoing maintenance.
The following table compares exosome and stem cell therapy across the key dimensions patients consider when choosing between them, including delivery time, cost, mechanism, and ideal use case.
| Factor | Exosome Therapy | Stem Cell Therapy |
|---|---|---|
| Delivery time | Same day (ready-to-use) | Approximately 7 weeks (culture period) |
| Cell culture required | No | Yes (autologous only) |
| Cost | Varies by application — contact us for details | Varies by condition — contact us for details |
| Best for | Mild conditions, maintenance, aesthetic enhancement, convenience | Severe/chronic conditions, structural damage, long-term regeneration |
| Mechanism | Paracrine signaling (molecular messages) | Cell integration + paracrine signaling |
| Frequency | Can repeat monthly or as needed | Typically 1–3 treatments over 1 year |
| Combination approach | Excellent for maintenance between stem cell treatments | Primary therapy, often followed by exosome maintenance |
As the comparison shows, exosomes are the “express option” for same-day results and ongoing maintenance, while stem cells deliver deeper regeneration for serious conditions. Many patients use both strategically — pursuing stem cell anti-aging therapy for primary treatment and maintaining results with periodic exosome sessions.
Reach us directly — WhatsApp and email inquiries are free of charge.
Who Is a Good Candidate for Exosome Therapy?
Exosome therapy is suitable for a broad range of patients seeking same-day regenerative improvement — from aesthetic enhancement and hair restoration to joint support and systemic wellness — though certain profiles benefit most from this approach.

You may not be a candidate if you have severe, longstanding joint degeneration that requires cell integration (advanced osteoarthritis may benefit more from stem cell therapy), extensive structural damage requiring cellular repair, active infection or severe immunosuppression, or if you are pregnant or nursing. Exosome therapy improves and regenerates — it does not replace stem cell therapy for the most advanced conditions.
Why Japan — and Why Cell Grand Clinic?
Japan operates under one of the world’s most advanced regulatory frameworks for regenerative medicine — the MHLW-administered Act on the Safety of Regenerative Medicine — making it a uniquely safe and transparent destination for exosome therapy backed by pharmaceutical-grade manufacturing.
Exosomes from a Stem Cell-Grade CPC Laboratory
Cell Grand Clinic’s exosomes are produced at the same Cell Processing Center (CPC) where the clinic’s stem cells are cultured — a facility built to pharmaceutical-grade Good Manufacturing Practice (GMP) standards. CPC-grade manufacturing delivers quality that general-purpose laboratories cannot match: verified exosome concentration (typically 100–200 million exosomes per mL, quantified by nanoparticle tracking analysis), rigorous characterization through size distribution and surface marker profiling (CD9, CD63, CD81), single-donor sourcing for consistency and traceability, and sterility and safety testing at every production lot.
Academic Collaboration with Prof. Takahiro Ochiya
The CPC laboratory maintains an academic collaboration with Professor Takahiro Ochiya, one of the world’s foremost authorities on extracellular vesicles and exosome biology. Prof. Ochiya currently serves at the Institute of Medical Science, Tokyo Medical University, and previously led the Division of Molecular and Cellular Medicine at the National Cancer Center Research Institute in Tokyo. He is a recipient of the Princess Takamatsu Cancer Research Prize — one of Japan’s most prestigious scientific honors — for his pioneering work on microRNA and extracellular vesicle signaling. This collaboration means the clinic’s exosome protocols are informed by decades of cutting-edge EV research.
Cell Grand Clinic: Physician-Led, Patient-Focused
Cell Grand Clinic, located in Shinsaibashi, Osaka, is an MHLW-certified regenerative medicine clinic with 13 distinct stem cell treatment plans filed and approved under Japan’s Act on the Safety of Regenerative Medicine. The clinic is directed by Dr. Yuichi Wakabayashi, M.D., Ph.D., who trained in regenerative medicine at the National Institutes of Health (NIH) and holds Diplomate status with the American Board of Regenerative Medicine (ABRM). Dr. Wakabayashi has administered over 3,000 stem cell treatments and brings rigorous, evidence-based practice standards to exosome therapy.
Ultra-High Concentration, Three Exosome Sources
One feature that sets Cell Grand Clinic apart is exosome concentration and source diversity. A single IV drip delivers approximately 300 billion exosomes — two vials of roughly 150 billion each — with concentration verified by ELISA assay (CD9, CD63, CD81 surface markers) at every production lot. That is several orders of magnitude above the doses used in many overseas clinics. In simple terms, more exosomes reaching your bloodstream means more signaling molecules available to support repair and regeneration throughout the body.
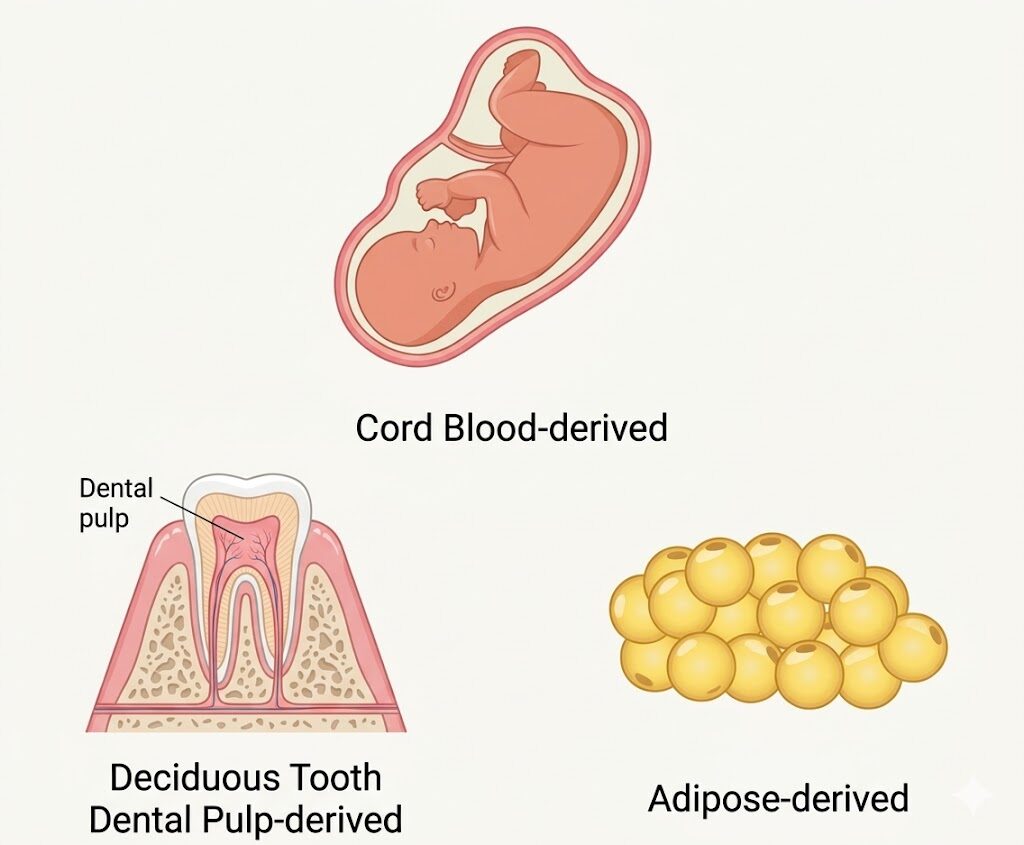
Cell Grand Clinic offers three distinct exosome sources, each derived from young Japanese donor stem cells (passage ≤ 3) cultured in serum-free media and freeze-dried for stability.
| Source | Key Growth Factor | Best Suited For |
|---|---|---|
| Umbilical Cord Blood | HGF-rich (hepatocyte growth factor) | Full-body tissue repair, anti-aging, skin rejuvenation |
| Dental Pulp | NGF-rich (nerve growth factor) | Nerve regeneration, cognitive support, diabetic neuropathy, ED |
| Adipose (Fat) | VEGF-rich (vascular endothelial growth factor) | Angiogenesis, hair regrowth (AGA), vascular aging |
The physician selects the optimal source — or a combination — based on the patient’s condition, target area, and treatment goals during consultation.
Think of it like choosing the right tool for the job: umbilical cord blood exosomes carry signals that tell the whole body to repair, dental pulp exosomes specialize in nerve-related healing, and adipose exosomes focus on building new blood vessels. Your physician matches the source to your specific needs.
The table below highlights how Cell Grand Clinic’s CPC-grade exosome manufacturing compares with typical providers in less-regulated markets. Key differences include production environment, quality verification, physician oversight, and academic backing.
| Factor | Cell Grand Clinic (Japan, MHLW-certified) | Mexico / Southeast Asia | Unregulated Markets |
|---|---|---|---|
| Government Oversight | MHLW certified ✓ | Limited or emerging regulation | No regulatory oversight |
| Production Facility | Pharmaceutical-grade CPC ✓ | Varies by clinic | Often unverified |
| Quality Verification | Per-lot NTA, marker profiling, functional assays ✓ | Variable quality controls | Rarely verified |
| Academic Backing | Prof. Ochiya (Tokyo Medical Univ.) ✓ | Typically no formal affiliation | No academic affiliation |
| Physician Credentials | NIH-trained MD/PhD; ABRM Diplomate ✓ | Varies; may include non-specialists | Often non-physician practitioners |
Comparison is based on general regulatory frameworks. Individual clinics may vary. Japan’s MHLW certification system (Act on the Safety of Regenerative Medicine) is unique in providing government-level oversight for regenerative treatments.
In summary, Cell Grand Clinic’s exosome therapy benefits from CPC-grade manufacturing, rigorous per-lot quality testing, academic collaboration with a leading EV researcher, and oversight by an NIH-trained regenerative medicine specialist — a combination that sets it apart from typical providers.
Reach us directly — WhatsApp and email inquiries are free of charge.
Frequently Asked Questions About Exosome Therapy
Q1: What is exosome therapy and how does it work?
Exosome therapy uses nano-sized vesicles from mesenchymal stem cells to deliver growth factors, microRNA, and anti-inflammatory signals that trigger your cells to repair and regenerate damaged or aging tissue through paracrine signaling.
Exosomes are extracellular vesicles (30–150 nanometers) released by mesenchymal stem cells. They carry the “repair instructions” of stem cells without being cells themselves. When injected into target tissue — skin, hair, joints, blood vessels — exosomes bind to cell surface receptors and transfer their cargo, signaling local cells to increase collagen, reduce inflammation, improve blood flow, and regenerate. This cell-to-cell communication through molecular messengers is called paracrine signaling.
Q2: Can I get exosome therapy on the same day as my visit?
Yes. Unlike stem cell therapy, which requires approximately 7 weeks of cell culture, exosome therapy uses pre-manufactured, high-concentration exosome preparations available immediately at the clinic.
You can arrive, complete a consultation, and receive treatment the same day. This makes exosome therapy ideal for travelers or anyone with a tight schedule. Results continue to develop over 2–4 weeks as your cells respond to the exosome signals.
Q3: Is exosome therapy safe?
Yes. A 2024 meta-analysis of 21 clinical trials found a serious adverse event rate of only 0.7% and a mild adverse event rate of 4.4% — comparable to or lower than other injectable treatments.
Serious adverse events requiring medical intervention were exceptionally rare. Minor adverse events — transient redness, mild swelling, minimal soreness — resolved without treatment within days. No deaths or permanent injuries were reported. Safety profiles were identical between autologous and allogeneic preparations.
Q4: How is exosome therapy different from stem cell therapy?
Stem cells are living cells that integrate and divide in your body; exosomes are their molecular messengers that deliver repair signals. Exosomes are faster and more convenient; stem cells are stronger for severe conditions.
Stem cell therapy transplants living mesenchymal stem cells that integrate, divide, and continuously release exosomes for months — ideal for severe, chronic conditions. Exosome therapy delivers only the signaling cargo immediately, with no culture period. Many patients combine both: stem cell therapy for primary treatment and exosome therapy for ongoing maintenance.
Q5: What areas of the body can be treated with exosomes?
Face, scalp, knee joints, penile tissue, and whole-body circulation via IV drip. An at-home nasal spray option is also available for cognitive and systemic support between clinic visits.
Facial injections improve wrinkles, texture, and tone. Scalp injections restore hair density. Knee injections provide cartilage support. Penile injections improve erectile function. IV infusions deliver exosomes systemically for whole-body anti-aging. Nasal spray bypasses the blood-brain barrier for CNS delivery.
Q6: How much does exosome therapy cost in Japan?
Pricing varies by application, exosome concentration, and number of sessions. Exosome therapy is generally more accessible than stem cell therapy — contact Cell Grand Clinic for a personalized estimate.
Costs depend on the treatment area (face, scalp, knee, IV, etc.) and the exosome concentration used. Most patients benefit from 1–3 treatments spaced 2–4 weeks apart. For a personalized estimate, reach out via WhatsApp or email — inquiries are free of charge.
Q7: Can I take exosome treatment home with me?
Yes. Intranasal exosome spray formulations are designed for at-home use, allowing you to continue regenerative benefits after your clinic visit in Japan.
Intranasal exosome spray is administered 2–3 times daily, 2–3 sprays per nostril. This non-invasive route, supported by 2026 research in the Journal of Controlled Release, reaches the CNS and provides systemic benefits. You can carry the spray with you and use it throughout the day — ideal for extending your treatment after returning home.
The Next Step: Regenerate While You’re Here
Exosome therapy represents a convergence of science, convenience, and proven results. Consistent skin improvement across 19 clinical studies. Hair density gains of up to 35 hairs/cm². A 0.7% serious adverse event rate. These are not marketing claims — they’re outcomes from peer-reviewed systematic reviews.
If you’re visiting Japan — whether for leisure, business, or wellness tourism — and you want visible regenerative improvement without a multi-week commitment, exosome therapy delivers. Same day. Evidence-based. Available now.
You don’t need to choose between time and results.
References
Flores Rodríguez J, Toledo Avelar L, Yi K, et al. Efficacy of exosome-based therapies for skin rejuvenation: a systematic review of human studies. Cureus. 2026;18(2):e104182. https://doi.org/10.7759/cureus.104182
Al Ameer MA, Alnajim AT, Al Ameer A, et al. Exosomes and hair regeneration: a systematic review of clinical evidence across alopecia types and exosome sources. Clin Cosmet Investig Dermatol. 2025;18:2215-2227. https://doi.org/10.2147/CCID.S543451
Van Delen S, et al. A systematic review and meta-analysis of clinical trials assessing safety and efficacy of human extracellular vesicle-based therapy. J Extracell Vesicles. 2024;13:e12458. https://doi.org/10.1002/jev2.12458
Wang X, et al. Stem cell-derived and plant-derived exosomes: Promising therapeutics for skin healing and regeneration. Mol Cell Probes. 2026;86:102064. https://doi.org/10.1016/j.mcp.2026.102064
Ayanji T, et al. Non-invasive administration of exosomes. J Control Release. 2026;392:114674. https://doi.org/10.1016/j.jconrel.2026.114674
Reach us directly — WhatsApp and email inquiries are free of charge.
最終更新日:2026.03.26



 Inquiries by Email
Inquiries by Email

