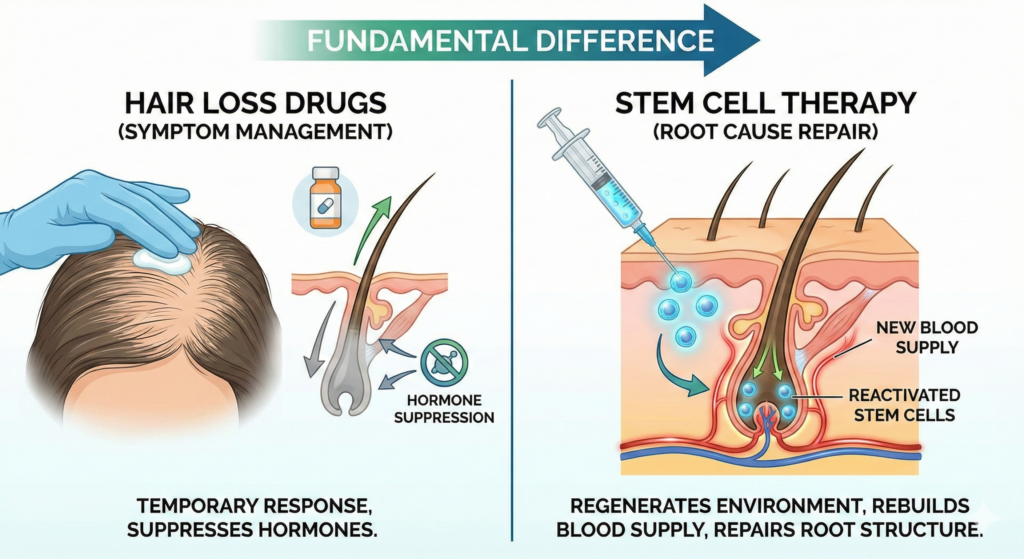
Hair loss isn’t vanity—it’s a signal that something has gone wrong beneath the scalp surface. Current treatments manage symptoms: finasteride blocks DHT (but carries sexual dysfunction and mood risks), minoxidil dilates blood vessels (but requires daily application forever), and hair transplants move existing hair from back to front (but can’t regenerate lost follicles and cost $15,000–$50,000). For women, finasteride is off-limits during pregnancy, and minoxidil yields inconsistent results.
What if there were a treatment that rebuilt the biological environment rather than just managing symptoms? Stem cell therapy works on a fundamentally different principle: instead of fighting DHT or coating the scalp with vasodilators, it regenerates the infrastructure—blood supply, growth factors, and the signaling environment that tells dormant follicles to grow again. This is not science fiction; it’s supported by randomized controlled trials, approved by Japan’s government, and available today.
- What Is Stem Cell Hair Restoration?
- What Are AGA and FAGA? Why Your Follicles Aren’t Dead
- How Stem Cell Therapy Reverses Hair Loss: The Science, Made Simple
- Stem Cells vs. Other Hair Loss Treatments
- Does Stem Cell Hair Restoration Work? Clinical Evidence
- Your Treatment at Cell Grand Clinic: Step by Step
- Why Japan — and Why Cell Grand Clinic?
- What to Expect: Your Results Timeline
- Who Is the Ideal Candidate?
- Frequently Asked Questions
- Closing: Why Now?
- References
What Is Stem Cell Hair Restoration?
Stem cell hair restoration uses a patient’s own fat-derived stem cells to reverse hair loss by rebuilding blood supply, reactivating dormant follicles, and repairing damaged structures. Learn more about how stem cell quality affects outcomes in our research library.
The Biology Behind Hair Loss
In male pattern hair loss (AGA), hair follicles shrink from thick, rooted structures to thin, superficial ones. In female pattern hair loss (FPHL), women lose density across the scalp. But here’s the critical discovery: the follicle stem cells aren’t dead.
Garza and colleagues at the University of Pennsylvania, published in the Journal of Clinical Investigation (2011), proved that even in bald scalps, follicle stem cells persist — the problem is in stem-to-progenitor conversion, not cell death. Matsumura et al. later showed in Science (2016) that COL17A1 deterioration causes stem cells to emigrate from follicles over time. This matters because dormant cells can be reactivated. Dead cells cannot.
What Are AGA and FAGA? Why Your Follicles Aren’t Dead
Androgenetic Alopecia (AGA) is genetically driven hair loss—commonly known as male pattern baldness. In women, it’s called Female Androgenetic Alopecia (FAGA) or female pattern hair loss. By age 50, roughly half of all men and nearly 40% of women experience noticeable thinning, and many notice changes as early as their late 20s.
Here’s what’s happening beneath the surface: dihydrotestosterone (DHT) binds to receptors in genetically susceptible follicles and gradually shrinks them. The growth phase (anagen) shortens cycle by cycle until each hair becomes too thin and short to see.
But here’s the critical point: in most AGA and FAGA cases, the follicle stem cells are still alive. They’re miniaturized and dormant—not dead. Research published in the Journal of Clinical Investigation confirmed that even in bald scalps, follicle stem cells remain present in the bulge region (Garza et al., 2011). They simply lack the right signals to reactivate.
This is precisely why stem cell therapy works—and why drugs that only block DHT can never fully reverse the damage.
How Stem Cell Therapy Reverses Hair Loss: The Science, Made Simple
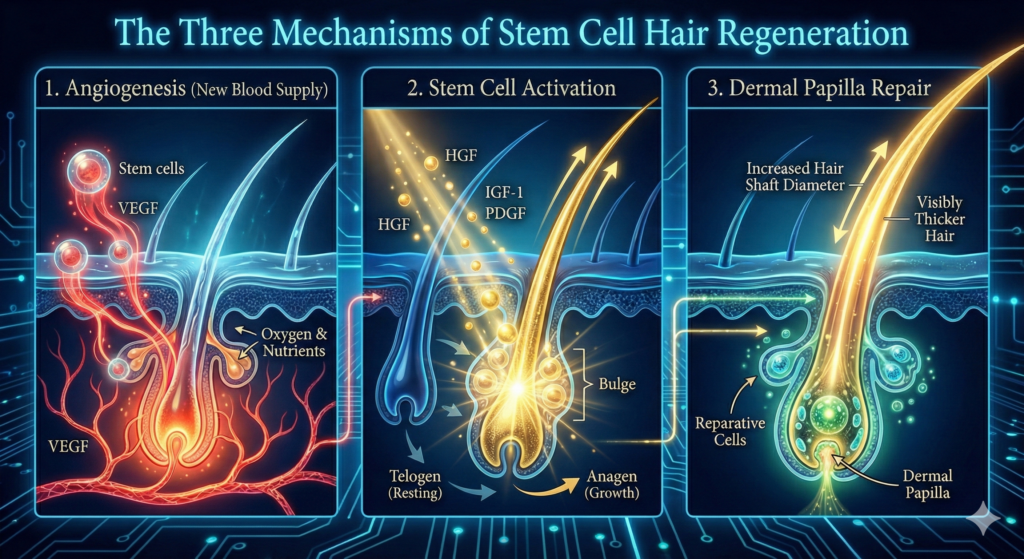
Stem cell therapy rebuilds the biological infrastructure damaged by hair loss. Three mechanisms:
Rebuilding Blood Supply (Angiogenesis)
Adipose-derived stem cells secrete VEGF (vascular endothelial growth factor), stimulating new blood vessels in the scalp. Yano et al. demonstrated in the Journal of Clinical Investigation (2001) that VEGF-mediated angiogenesis directly controls follicle size and hair growth — in plain terms, more blood supply means bigger, healthier follicles.
Reactivating Dormant Follicles
Stem cells secrete growth factors (HGF, IGF-1, PDGF) that reactivate dormant follicles and extend the growth phase. The signals stem cells produce drive recovery.
Repairing the Dermal Papilla
The dermal papilla “commands” hair growth. In hair loss, it shrinks. Stem cells rebuild it. Thicker dermal papillas = thicker hair growth.
Stem Cells vs. Other Hair Loss Treatments
Comparison Table: How Treatments Differ
| Mechanism | Finasteride | Minoxidil | Hair Transplant | PRP | Stem Cell Therapy |
|---|---|---|---|---|---|
| How It Works | Blocks DHT | Vasodilator | Surgical transplant | Platelet concentrate | Regenerates follicle infrastructure |
| Duration of Results | Requires ongoing use | Requires ongoing use | Permanent | Months–1 year (requires repeat) | 1–3+ years reported |
| Common Side Effects | Sexual dysfunction, mood, breast tissue changes | Scalp irritation, facial hair growth | Surgical complication (scarring, infection) | Scalp pain, swelling | Minimal (temporary irritation) |
| Safe for Women? | No (pregnancy risk) | Yes, but weak results | Yes | Yes | Yes |
| Actually Repairs Follicles? | No (suppresses only) | No (vasodilation only) | No (transplants, doesn’t repair) | Partial | Yes (regenerative) |
| Typical Cost Range | $120–240/year | $200–300/year | $15,000–50,000 | $500–2,000 per session | $13,000–27,000 (one-time) |
Why Stem Cells Stand Apart
Finasteride and minoxidil manage symptoms but require forever use; hair loss accelerates when stopped. Hair transplants move existing hair but can’t regenerate lost follicles. Stem cell therapy targets the root cause, restoring the biological environment. Early-stage loss can be reversed; advanced loss can be stabilized. Crucially, women can use it without pregnancy concerns.
Does Stem Cell Hair Restoration Work? Clinical Evidence
Yes. Multiple randomized controlled trials and a systematic review of 12 RCTs confirm that adipose-derived stem cell therapies produce statistically significant increases in hair count and density, with zero serious adverse effects reported across all published trials.
The evidence isn’t theoretical—it’s in peer-reviewed medical journals.
Evidence Table: What Clinical Trials Show
Clinical trials demonstrate consistent improvements in hair density and count across multiple patient populations. The table below reflects published, peer-reviewed evidence from randomized controlled trials and systematic reviews.
| Study | Year | Design | Patients (n) | Key Finding | Follow-up |
|---|---|---|---|---|---|
| Gentile Int J Mol Sci | 2019 | Controlled, half-head (micrografts) | 33 | 33% ± 7.5% hair density increase; follicle density 0.46→1.4/mm² | 23 wk; sustained 11 mo |
| Tak et al. Stem Cells Transl Med | 2020 | RCT, double-blind (ADSC extract, topical) | 34 | 28.1% hair count increase; 14.2% diameter increase vs 7.1% control | 16 wk |
| Gasteratos et al. Plast Reconstr Surg Glob Open | 2024 | Systematic review (12 RCTs) | 514 | Confirmed safety and efficacy; zero serious adverse effects across all trials | 2013–2023 |
| Moneib et al. Int J Trichology | 2025 | RCT, women only (ADSCs injected) | 33 | Improved density, thickness, follicular units; ↑follistatin, ↓DKK-1 | 12 & 24 wk |
| Shin et al. Int J Dermatol | 2015 | Retrospective (ADSC conditioned media) | 27 | 16.4% hair density increase (105.4→122.7/cm²); thickness ↑ | 12 wk |
All studies used human subjects. Full references with DOI links are provided at the end of this article.
Key studies include Gentile et al. (2019) published in Int J Mol Sci, Tak et al. (2020) in Stem Cells Transl Med, Gasteratos et al. (2024) in Plast Reconstr Surg Glob Open, Moneib et al. (2025) in Int J Trichology, and Shin et al. (2015) in Int J Dermatol.
Across all five studies, adipose-derived stem cell therapies produce measurable improvements in hair density and count, with zero serious adverse effects across 500+ patients. The Moneib 2025 trial directly used cultured autologous ADSCs injected into the scalp, while earlier studies used extracts or conditioned media — an important distinction when evaluating how different protocols may apply to your case.
What the Numbers Mean
Gentile 2019: Follicle density rose from 0.46 to 1.4 per mm²—three times more follicles per mm². Results vary (20–35%) by baseline, genetics, and duration. Zero serious adverse effects across 514 patients vs. 5–15% complications with transplants.
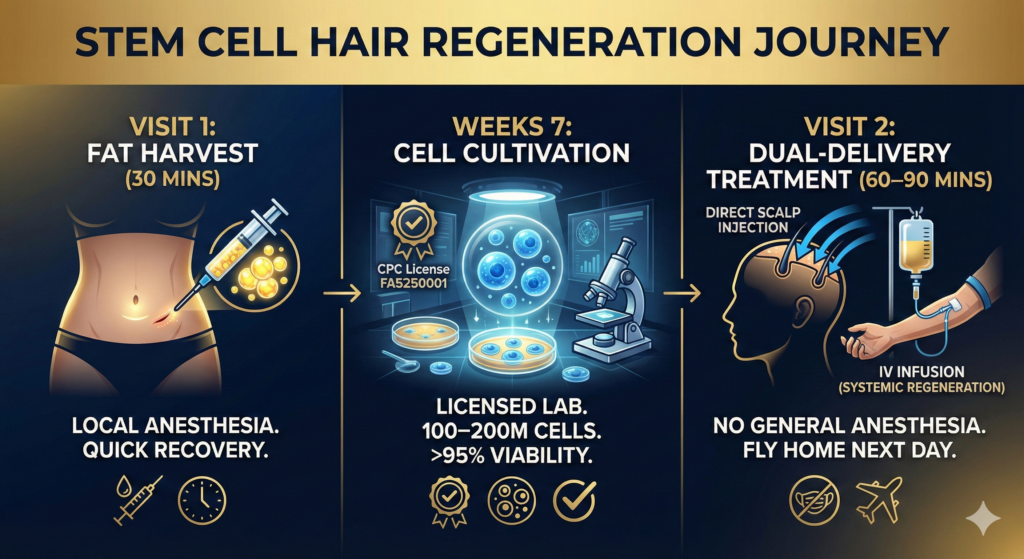
Your Treatment at Cell Grand Clinic: Step by Step
Our protocol is designed specifically for international patients. The entire process requires just two visits to Osaka, approximately 2–3 weeks apart.
Visit 1: Fat Harvest (30 Minutes)
Under local anesthesia, approximately 10ml of fat is extracted from your abdomen through a small incision. You can resume normal activities the same day.
Weeks 7: Cell Cultivation in Our Licensed Laboratory
Your tissue is transported to our government-certified Cell Processing Center (CPC License: FA5250001), where stem cells are isolated and cultured to reach 100 to 200 million cells with over 95% viability. Every batch is tested for sterility, viability, and potency before treatment.
Visit 2: Dual-Delivery Treatment Day
Direct Scalp Injection delivers stem cells precisely into the dermal papilla layer—exactly where follicle regeneration needs to happen. This targeted approach maximizes concentration where it matters most. IV Infusion (optional but recommended) delivers stem cells systemically, supporting vascular health and overall regeneration. Many patients combine scalp treatment with broader anti-aging benefits. Total treatment time: approximately 60–90 minutes. No general anesthesia. No hospitalization. Fly home the next day.
Reach us directly — WhatsApp and email inquiries are free of charge.
Why Japan — and Why Cell Grand Clinic?
Japan’s Regulatory Advantage
Japan is the only major country with a dedicated national law for regenerative medicine. The Act on the Safety of Regenerative Medicine (2014) requires every stem cell treatment to be government-approved before patient administration, processed in licensed pharmaceutical-grade facilities, and reported to a national safety database for ongoing surveillance. This means every treatment we provide is tracked, audited, and subject to national-level quality control. Many stem cell tourism destinations in Southeast Asia, Central America, and Eastern Europe operate without comparable oversight—lower prices often reflect absent safety standards, not better value.
Cell Grand Clinic holds 10 Type II Government Licenses — the highest outpatient classification issued by Japan’s Ministry of Health, Labour and Welfare.Why Japan—and Why Cell Grand Clinic

World-Class Scientific Partnership
Your stem cells are cultured at a government-certified Cell Processing Center (CPC License: FA5250001) connected to the research of Professor Takahiro Ochiya of Tokyo Medical University — a Web of Science Top 0.1% Highly Cited Researcher for six consecutive years, with over 800 peer-reviewed publications and 57,000+ citations. His pioneering exosome research directly informs the cultivation protocols used on your cells. This caliber of academic-clinical partnership is virtually unheard of in the private regenerative medicine sector anywhere in the world.
Cell Quality Standard: Fresh, Pure, Young
Many clinics offer a few million cells—doses too small for systemic effect. We invest 7 weeks cultivating your cells to a maximum of 200 million viable cells (2×10⁸). Every batch is over 95% survival rate, only early-passage (P3) cells for maximum potency, tested for sterility and genetic stability. Our philosophy: “Fresh, Pure, Young”—
| Quality Pillar | Our Standard | Why It Matters |
| FRESH | 95%+ cell survival rate | Higher survival rate = greater regenerative and repair potential. Dead or dying cells cannot heal. Cell Viability ≥95% (verified before every administration) vs. industry average of 70–85% |
| PURE | ~99% stem cell surface antigen expression | CD73+, CD90+, CD105+ expression ≈99%; CD45−, CD34− (contaminants eliminated) vs. many clinics that do not test |
| YOUNG | Passage 3 cultivation; never over-expanded | Early-passage cells maintain maximum differentiation ability and biological activity. Over-passaged cells lose potency. Guaranteed 100 million cells at P3 vs. competitors using P5+ with reduced potency |
| GUARANTEED DOSE | 100 to 200 million cells per administration guaranteed | Therapeutic dosing matters. Insufficient cell counts mean insufficient results. |

Your Doctor: A Physician-Scientist Who Speaks Your Language
Dr. Yuichi Wakabayashi, M.D., Ph.D. brings credentials that bridge East and West. He completed postdoctoral training at the National Institutes of Health (NIH) in the United States. He is the first author on a Pfizer-collaborated world-first PET tracer study published in the Journal of Nuclear Medicine (2022), a Diplomate of the American Board of Regenerative Medicine (ABRM), and was featured in The Wall Street Journal as a “Next Era Leader.” Critically, Dr. Wakabayashi is fluent in English, allowing direct doctor-patient communication with no interpreters needed.
What to Expect: Your Results Timeline
| Timeframe | What to Expect |
| Weeks 1–4 | Reduced shedding; early signs of follicle reactivation beginning at the cellular level. |
| Weeks 4–12 | New fine hairs (vellus) emerging; existing hairs may begin to feel thicker. |
| Months 3–6 | Visible improvement in density and thickness. Clinical trials show statistically significant results by week 8–16. |
| Months 6–12 | Peak results—fuller, thicker, stronger hair. Many patients report the improvement continues beyond the 6-month mark. |
| 12+ Months | Sustained results. Annual maintenance treatment recommended for long-term optimization. |
Individual results vary depending on the degree of hair loss, overall health, and genetics. Patients with earlier-stage AGA/FAGA typically see the most dramatic improvements—another reason not to delay.
Who Is the Ideal Candidate?
Stem cell hair restoration works best for certain patients. Here’s how to assess your candidacy.
Norwood-Hamilton I–V or Ludwig I–II classification
Rather than decades-long stable baldness
Even if miniaturized; these can be reactivated
Stem cell therapy is gradual and natural-looking
Logistics can be coordinated with the clinic
These must be managed before treatment
Very few follicles remain to reactivate
Scarred follicles cannot regenerate; new follicles cannot grow in scar tissue
These require nutritional correction first; stem cells help but aren’t primary treatment
Stem cells help but require dermatology co-management and immune-suppressing therapies
Realistic Expectations
If you’re a good candidate, here’s what to expect realistically:
- Hair density increase of 15–35% (not 100% regrowth of lost hair)
- Thicker, healthier-looking hair even if density increase is modest
- Stabilized hair loss (no further shedding acceleration)
- Results visible after 3–4 months, peak by 6–12 months
- Duration: 1–3+ years reported (varies individually; some patients may choose repeat treatments)
Individual results vary. Some patients see dramatic improvements. Others see modest but meaningful improvements. Genetics, age, and duration of hair loss all influence the outcome.
Frequently Asked Questions
Q1: Does Stem Cell Hair Restoration Actually Work?
Yes. Randomized controlled trials show 33% hair density increase across 514 patients reviewed in a 2024 systematic analysis, with zero serious adverse effects. Results vary by individual hair loss pattern and severity.
Q2: How Much Does Stem Cell Hair Restoration Cost?
¥2–4 million JPY ($13,000–$27,000 USD) depending on cell count and delivery method. Cost varies by cell count (100M vs. 200M), delivery method (injection alone vs. injection + IV), and testing packages; transparent pricing is provided at consultation.
Q3: What Exactly Is Stem Cell Hair Restoration?
A procedure using your own fat-derived stem cells to regenerate the biological environment that supports hair growth. Stem cells are harvested from fat tissue, cultured for 7 weeks, then injected into the scalp. The cells migrate to follicles, secreting growth factors that reactivate dormant follicles and rebuild the dermal papilla.
Q4: Is Stem Cell Therapy for Hair Loss Safe?
Yes. Zero serious adverse effects were reported across 514 patients in a 2024 systematic review. Temporary effects (redness, swelling, tenderness) resolve within 3–7 days. No infections, scarring, or nerve injury have been documented. Because the cells are autologous, there’s no immune rejection risk.
Q5: Can Women Receive Stem Cell Hair Restoration?
Yes. A 2025 RCT of 33 women showed improved hair density, thickness, and follicular unit numbers. Stem cell therapy doesn’t suppress hormones or interfere with pregnancy planning, unlike finasteride.
Q6: How Does Stem Cell Therapy Compare to Hair Transplant?
Transplants move hair (permanent, expensive). Stem cells regenerate follicles (non-surgical, slower). Combine both for optimal results.
Q7: How Long Do Stem Cell Hair Restoration Results Last?
1–3+ years, longer than PRP. Genetics, age, and scalp health influence stability. Hygiene and nutrition help maintain results.
Q8: I Live Outside Japan. How Does the Process Work?
Option 1: Local harvest, Cell Grand cultivates cells, you receive injection locally. Option 2: Travel to Osaka (two visits, ~3 weeks apart).
Q9: Can I Combine Stem Cell Therapy With Other Hair Loss Treatments?
Yes. Finasteride (DHT suppression) + stem cells (repair) work synergistically. Discuss with your physician.
Closing: Why Now?
Hair loss is progressive. The longer you wait, the more follicles miniaturize past the point of easy reactivation. A follicle that’s been dormant for 2 years is easier to reawaken than one dormant for 10 years—and much easier to save before it becomes dormant at all.
If you’re in early-to-moderate hair loss and considering stem cell therapy, the time to act is now. Not impulsively—but thoughtfully, with a consultation, with full information, with realistic expectations.
Finasteride and minoxidil are management tools. Hair transplant is a localized solution. Stem cell therapy is the only treatment that addresses the biological cause of hair loss—and it works non-surgically, without daily medication, for both men and women. Learn more about Cell Grand Clinic’s approach.
Reach us directly — WhatsApp and email inquiries are free of charge. Our English-speaking team will review your photos, assess your candidacy, and explain your options.
References
Garza, L. A., Yang, C. C., Zhao, T., Blatt, H. B., Lee, M., He, H., … Cotsarelis, G. (2011). Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. Journal of Clinical Investigation, 121(2), 613–622. https://doi.org/10.1172/JCI44478
Matsumura, H., Mohri, Y., Binh, N. T., Tateishi, T., Harada, M., Nakamura, Y., … Nishimura, E. K. (2016). Hair follicle aging is driven by transepidermal elimination of stem cells via COL17A1 proteolysis. Science, 351(6273), aad4395. https://doi.org/10.1126/science.aad4395
Gentile, P. (2019). Autologous cellular method using micrografts of human adipose tissue derived follicle stem cells in androgenic alopecia. International Journal of Molecular Sciences, 20(14), 3446. https://doi.org/10.3390/ijms20143446
Tak, Y. J., Lee, S. Y., Cho, A. R., & Kim, Y. S. (2020). A randomized, double-blind, vehicle-controlled clinical study of hair regeneration using adipose-derived stem cell constituent extract in androgenetic alopecia. Stem Cells Translational Medicine, 9(8), 839–849. https://doi.org/10.1002/sctm.19-0410
Gasteratos, K., Kouzounis, K., & Goverman, J. (2024). Autologous stem cell-derived therapies for androgenetic alopecia: A systematic review of randomized control trials on efficacy, safety, and outcomes. Plastic and Reconstructive Surgery Global Open, 12(2), e5606. https://doi.org/10.1097/GOX.0000000000005606
Moneib, H., Fathy, G., Samir, N. A., El-Khazragy, N., & El-Bassiouny, M. (2025). Successful treatment of female pattern hair loss with injection of autologous adipose-derived adult stem cells. International Journal of Trichology, 17(2), 113–120. https://doi.org/10.4103/ijt.ijt_129_23
Shin, H., Ryu, H. H., Kwon, O., Park, B. S., & Jo, S. J. (2015). Clinical use of conditioned media of adipose tissue-derived stem cells in female pattern hair loss: A retrospective case series study. International Journal of Dermatology, 54(6), 730–735. https://doi.org/10.1111/ijd.12650
Yano, K., Brown, L. F., & Detmar, M. (2001). Control of hair growth and follicle size by VEGF-mediated angiogenesis. Journal of Clinical Investigation, 107(4), 409–417. https://doi.org/10.1172/JCI11317
Zuk, P. A., Zhu, M., Mizuno, H., Huang, J., Futrell, J. W., Katz, A. J., … Hedrick, M. H. (2002). Multilineage cells from human adipose tissue: Implications for cell-based therapies and regenerative medicine. Molecular Biology of the Cell, 13(12), 4279–4295. https://doi.org/10.1091/mbc.e02-02-0105
最終更新日:2026.03.26



 Inquiries by Email
Inquiries by Email

