- Opening
- What Is Neuropathy — and Why Conventional Treatments Fall Short
- Types of Neuropathy That May Respond to Stem Cell Therapy
- How Stem Cells May Repair Damaged Nerves: The Science
- Clinical Evidence: What the Research Shows
- Who May Benefit — and What to Expect
- Why Japan — and Why Cell Grand Clinic?
- Frequently Asked Questions
- Take the Next Step
- References
Opening
Your hands tingle at night. Your feet feel numb, as if wrapped in invisible gauze. Medications take the edge off — but never truly restore what was lost. If peripheral neuropathy has stolen your sensation, your balance, or your sleep, you are not alone — and conventional medicine may have already reached its limits for you.
Emerging clinical research suggests that mesenchymal stem cell (MSC) therapy may offer a regenerative path forward — one that targets the underlying nerve damage rather than merely masking symptoms.

What Is Neuropathy — and Why Conventional Treatments Fall Short
Neuropathy is a condition in which peripheral nerves — the network carrying signals between the brain, spinal cord, and the rest of the body — become damaged, leading to pain, numbness, and weakness most often in the hands and feet. Mesenchymal stem cell therapy has emerged as a regenerative approach that may help repair this nerve damage through paracrine signaling, immunomodulation, and neurotrophic factor secretion.
A comprehensive 2026 review published in JAMA reports that peripheral neuropathy affects roughly 1% of the general adult population worldwide, with prevalence rising sharply among older adults and those with chronic conditions. Diabetes is the single most common cause: more than 206 million people globally live with diabetes, and over 50% of peripheral neuropathy cases in Western populations are diabetic in origin. Another 27% of cases remain idiopathic — meaning no identifiable cause is ever found.
Despite these staggering numbers, treatment options remain frustratingly limited. The same JAMA review notes that gabapentin, one of the most widely prescribed medications for neuropathic pain, achieves a 50% or greater reduction in pain intensity in only 38% of patients with diabetic peripheral neuropathy. Many patients cycle through multiple drugs — pregabalin, duloxetine, tricyclic antidepressants — searching for relief that may never fully arrive.
Types of Neuropathy That May Respond to Stem Cell Therapy
Not all neuropathy is the same. The potential for stem cell therapy varies by type, stage, and underlying cause. Based on the current body of research, several forms stand out as particularly relevant.
Diabetic Peripheral Neuropathy (DPN)
Diabetic peripheral neuropathy is the most extensively studied form in the context of stem cell therapy. It typically begins in the feet and progresses upward, causing burning pain, numbness, and loss of protective sensation that can lead to ulcers and amputation. According to the 2026 JAMA review, DPN develops in a substantial proportion of diabetic patients and is the leading cause of non-traumatic lower limb amputation worldwide. Current pharmacological treatments manage symptoms but do not reverse the underlying nerve fiber loss.
Chemotherapy-Induced Peripheral Neuropathy (CIPN)
Cancer survivors frequently develop peripheral neuropathy as a side effect of platinum-based, taxane, or vinca alkaloid chemotherapy agents. CIPN can persist for months or years after treatment ends, affecting up to 68% of patients in the first month and remaining chronic in approximately 30%. No FDA-approved treatment currently exists specifically for CIPN, making it a condition of significant unmet need.
Small Fiber Neuropathy and Idiopathic Neuropathy
Small fiber neuropathy selectively damages the thin, unmyelinated nerve fibers responsible for pain and temperature sensation. It causes burning pain, often in a “stocking-and-glove” distribution, and is notoriously difficult to diagnose with standard nerve conduction studies. For the 27% of neuropathy patients whose condition is classified as idiopathic — no known cause — treatment options are especially limited, as there is no underlying disease to target.
How Stem Cells May Repair Damaged Nerves: The Science
A natural question arises: how could stem cells — typically associated with orthopedic or cosmetic applications — help with nerve damage? The answer lies not in the cells themselves replacing lost neurons, but in their role as biological signal centers.
A 2025 review published in Frontiers in Cell and Developmental Biology provides a comprehensive overview of how mesenchymal stem cells (MSCs) interact with damaged peripheral nerves. The mechanisms are multifaceted.
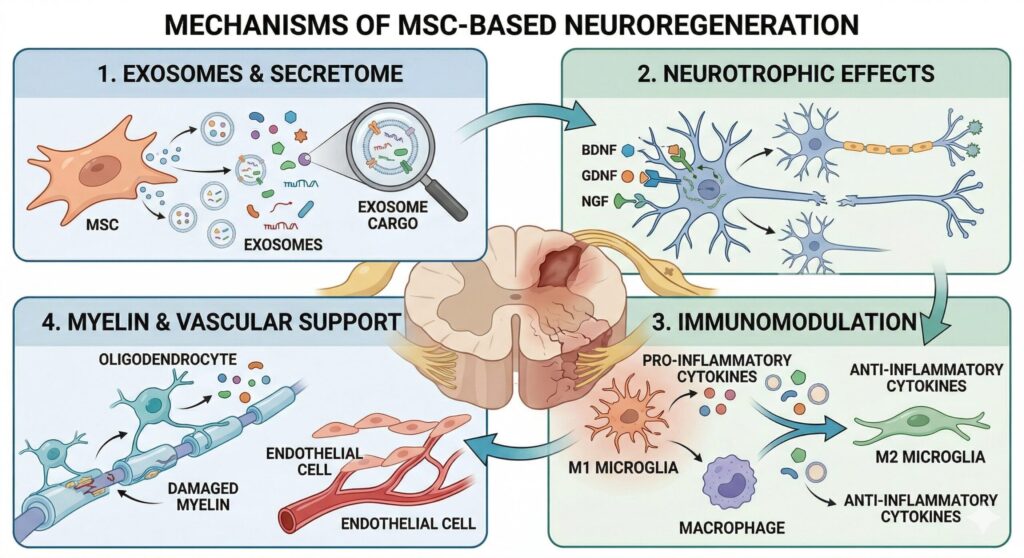
Neurotrophic factor secretion. MSCs release brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) — two proteins that are critical for the survival, growth, and repair of neurons. These factors provide a neuroprotective environment around damaged nerve fibers, supporting regeneration rather than further degeneration.
Immunomodulation. Chronic neuropathy is often accompanied by persistent neuroinflammation. MSCs can shift the immune environment from a pro-inflammatory state to a reparative one — specifically by inhibiting M1-type macrophages (which drive inflammation) and promoting M2-type macrophages (which support tissue repair). The review identifies suppression of NF-κB and PI3K/AKT signaling pathways as key mechanisms behind this shift.
Exosomes and secretome. Beyond direct cell-to-cell contact, MSCs release tiny vesicles called exosomes that carry bioactive molecules — cytokines, growth factors, and microRNAs — to distant tissues. These acellular components have demonstrated anti-inflammatory, neuroprotective, and immunomodulatory properties in their own right, suggesting that even the “byproducts” of stem cells may contribute to nerve healing.
Myelin and vascular support. Peripheral nerves depend on intact myelin sheaths for signal conduction and on healthy blood supply for oxygen and nutrients. MSCs have been shown to promote both remyelination and angiogenesis (new blood vessel formation), addressing two fundamental requirements for nerve recovery.
Clinical Evidence: What the Research Shows
The scientific rationale is compelling — but does it translate into measurable improvements for real patients? A growing body of clinical evidence — including a randomized controlled trial with 8 years of follow-up, two systematic reviews, and multiple controlled trials — offers encouraging data.
Nerve Conduction Velocity: Objective Evidence of Repair
A 2024 systematic review and meta-analysis published in Stem Cell Research & Therapy pooled data from 7 controlled clinical trials — selected from an initial screening of 5,431 studies — focusing exclusively on human patients with diabetic peripheral neuropathy. The stem cell types used in these trials were bone marrow-derived mononuclear cells (BM-MNCs) and umbilical cord-derived mesenchymal stem cells (UC-MSCs), administered primarily through intramuscular injection.
The results were statistically significant across multiple endpoints. Motor nerve conduction velocity improved by a weighted mean difference (WMD) of 2.2 m/s (95% CI: 1.6–2.8) — in practical terms, this means that the electrical signals traveling through motor nerves were measurably faster after stem cell treatment, indicating genuine nerve fiber repair or regeneration. Sensory nerve conduction velocity also improved, with a WMD of 1.9 m/s (95% CI: 1.1–2.6), suggesting that the nerves responsible for touch and temperature sensation were also recovering.
Clinical symptom scores reinforced these objective findings. The Toronto Clinical Scoring System (TCSS), a validated composite measure of neuropathy severity, improved by a WMD of −3.6 points (95% CI: −5.0 to −2.2) — a meaningful reduction in symptom burden that patients could feel in their daily lives. Vibration perception threshold, another objective marker, improved by a WMD of −2.9 (95% CI: −4.0 to −1.8).
Importantly, complications were limited to injection-site pain and mild swelling — no serious adverse events were attributed to the stem cell treatment itself.
It is worth noting that these trials used BM-MNCs and UC-MSCs, not adipose-derived stem cells (ADSCs). However, the underlying paracrine and regenerative mechanisms — neurotrophic factor secretion, immunomodulation, exosome release — are shared across mesenchymal stem cell types, suggesting comparable therapeutic potential regardless of tissue source.
Adipose Tissue-Derived Components: Direct Relevance to ADSC Therapy
A 2024 systematic review published in Bioengineering specifically examined human clinical studies of adipose tissue-derived components — including fat grafting and stromal vascular fraction (SVF) — for various forms of neuropathic pain. The review analyzed 10 clinical studies identified from 433 initial records, covering indications such as post-mastectomy pain syndrome, neuromas, post-herpetic neuropathy, neuropathic scar pain, and trigeminal neuropathic pain.
In 7 of the 10 studies, neuropathic pain levels decreased following treatment. While the authors note that overall quality of life did not consistently improve across all studies, and that larger randomized controlled trials are needed, the finding that adipose-derived treatments reduced neuropathic pain in the majority of clinical settings is particularly relevant — as it demonstrates that the specific tissue source used in ADSC-based regenerative protocols has a direct clinical track record in nerve pain conditions.
Long-Term Prevention: An 8-Year Randomized Controlled Trial
Perhaps the most striking evidence comes from a 2024 randomized controlled trial published in Stem Cell Research & Therapy that followed 97 patients with type 2 diabetes for 8 years — making it one of the longest-running stem cell trials in the diabetes and neuropathy space. Patients were randomized into three groups: bone marrow MSCs combined with mononuclear cells (n=33), mononuclear cells alone (n=32), or standard medical treatment only (n=31). The trial was registered with ClinicalTrials.gov (NCT01719640).
At the 8-year mark, the results were dramatic. Only 10.3% of patients in the MSC combination group developed diabetic peripheral neuropathy, compared to 48.3% in the control group (p=0.0015) — meaning stem cell-treated patients were nearly five times less likely to develop neuropathy over eight years. Even the mononuclear cell-only group showed significant protection, with a DPN incidence of 17.9% (p=0.015 vs. control). Adverse events were limited to mild, transient symptoms such as abdominal discomfort and brief fever — no serious safety concerns emerged over the entire 8-year observation period.
This trial used bone marrow-derived MSCs and mononuclear cells delivered via intraarterial and intravenous routes, which differs from the adipose-derived stem cell protocol used by many clinics today. However, the core regenerative and anti-inflammatory mechanisms are shared across MSC types, and the study’s demonstration that stem cell therapy can prevent long-term nerve damage in diabetic patients represents a significant milestone.
The following table summarizes the key evidence across all three clinical studies.
| Study | Year | Design | Trials (n) | Key Finding | Cell Type |
|---|---|---|---|---|---|
| Alizadeh et al. Stem Cell Res Ther | 2024 | SR & Meta-analysis | 7 | Motor NCV +2.2 m/s, Sensory NCV +1.9 m/s, TCSS −3.6 points | BM-MNC, UC-MSC |
| Wu et al. Stem Cell Res Ther | 2024 | RCT, 8-year follow-up | 97 | DPN incidence: 10.3% (MSC) vs 48.3% (control), p=0.0015 | BM-MSC + BM-MNC |
| Claessens et al. Bioengineering | 2024 | Systematic Review | 10 | 7/10 studies showed neuropathic pain reduction | Fat graft, SVF (adipose-derived) |
All studies used human subjects. Full references with DOI links are provided at the end of this article.
Who May Benefit — and What to Expect
Stem cell therapy for neuropathy is not a universal solution, and not every patient will be an ideal candidate. Based on the clinical evidence and Cell Grand Clinic’s experience treating over 3,000 patients with various conditions, the following profile describes individuals who may benefit most from this regenerative approach.
Individual suitability is determined during a medical consultation. This checklist is for general reference only.
The treatment process at Cell Grand Clinic involves two visits to Osaka, separated by approximately seven weeks. During the first visit, a small amount of adipose (fat) tissue is collected from the abdomen under local anesthesia — a procedure that takes about 20 minutes. Over the following seven weeks, stem cells are isolated and expanded in a CPC-grade (Cell Processing Center) laboratory to reach up to 200 million cells. During the second visit, the cultured cells are administered via intravenous infusion, allowing them to circulate throughout the body and reach damaged nerve tissues.
Results are not immediate. Most patients begin noticing changes gradually over a period of weeks to months, as the regenerative and anti-inflammatory processes unfold. Some patients choose to undergo a second course of treatment to reinforce the initial response. For an overview of all conditions treated with stem cell therapy at Cell Grand Clinic, visit the clinic’s English homepage.
Why Japan — and Why Cell Grand Clinic?
If you are considering stem cell therapy for neuropathy, the choice of where to receive treatment matters as much as the treatment itself. Stem cell clinics operate in dozens of countries, but regulatory frameworks vary enormously — and the difference can determine both your safety and your results.
Japan stands apart for one critical reason: it is the only major nation with a dedicated legal framework governing regenerative medicine. The Act on the Safety of Regenerative Medicine, enforced by the Ministry of Health, Labour and Welfare (MHLW), requires every clinic offering stem cell treatments to register each therapy plan with the government, undergo review by independent certified committees, and maintain CPC-grade laboratories that meet strict manufacturing standards. This is not voluntary accreditation — it is law.
Cell Grand Clinic Corabolates this framework with CPC License FA5250001, under the medical direction of M.D., Ph.D. Yuichi Wakabayashi, who completed postdoctoral training at the National Institutes of Health (NIH) in the United States and holds board certification from the American Board of Regenerative Medicine (ABRM).

Grand Stem Cell: Four Quality Standards
What sets Cell Grand Clinic’s stem cells apart is a commitment to four non-negotiable quality benchmarks that define every Grand Stem Cell treatment:
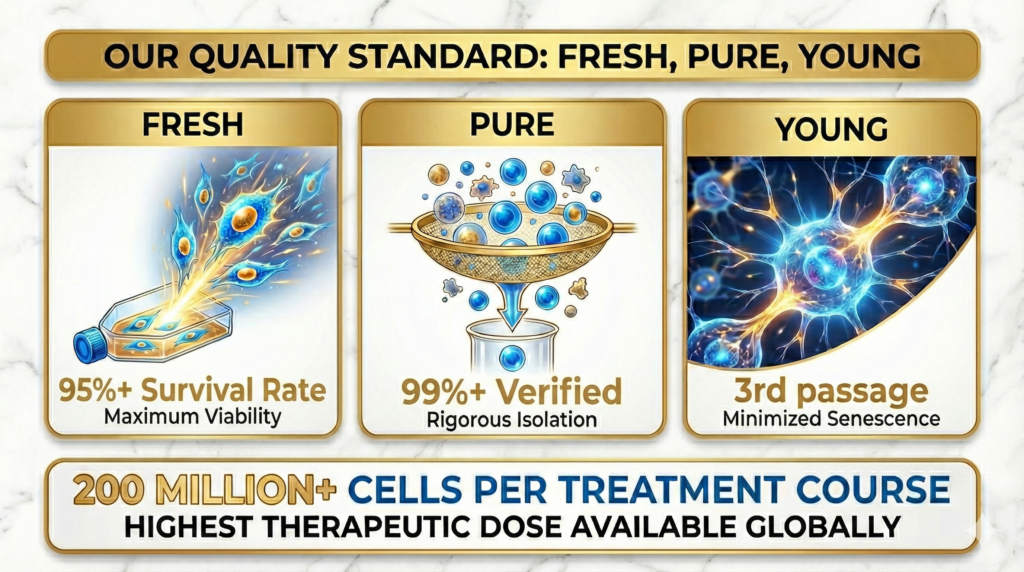
Cultured Exclusively for You. Grand Stem Cells are never off-the-shelf or shared. After your treatment plan is decided, cells are cultured exclusively from your own adipose tissue over a 7-week period. Every cell is grown for one patient and one patient only.
ISCT-Standard Verification. Surface marker testing follows the International Society for Cellular Therapy (ISCT) global standard, confirming that every administered cell is a genuine mesenchymal stem cell. Cells that do not meet criteria are discarded.
95%+ Viability — Only Living Cells. Dead or degraded cells cannot contribute to tissue repair. Cell Grand Clinic verifies that viability remains above 95% right up to the moment of administration, maintaining quality control through the final step.
Youth and Volume, Without Compromise. Stem cells age with repeated culturing, losing regenerative capacity. Grand Stem Cells are strictly limited to Passage 3 or below, and up to 200 million cells are cultured while preserving their youthful activity and potency.
Every patient receives a Certificate of Quality documenting that their Grand Stem Cells have met all four standards.
The following table illustrates how Japan’s regulatory environment compares with popular stem cell tourism destinations.
| Factor | Japan (MHLW-certified) | Mexico | Thailand |
|---|---|---|---|
| Government Oversight | MHLW certified ✓ | Limited regulation | Emerging regulation |
| Cell Quality Standards | CPC-grade, 7-week culture ✓ | Varies by clinic | Varies by clinic |
| Cell Count per Treatment | Up to 200 million ✓ | Varies | Varies |
| Autologous (Own Cells) | Yes ✓ | Often allogeneic | Mixed |
| Physician Credentials | NIH-trained MD ✓ | Varies | Varies |
Comparison is based on general regulatory frameworks. Individual clinics may vary. Japan’s MHLW certification system (Act on the Safety of Regenerative Medicine) is unique in providing government-level oversight for regenerative treatments.
Reach us directly — WhatsApp and email inquiries are free of charge.
Frequently Asked Questions
Can stem cells cure neuropathy?
Current evidence suggests that stem cell therapy may help improve nerve function and reduce neuropathic symptoms, but it is not considered a cure. Clinical trials have demonstrated measurable improvements in nerve conduction velocity and symptom scores in patients with diabetic peripheral neuropathy. However, the degree of improvement varies by individual, the type and severity of neuropathy, and how early treatment is initiated. Stem cell therapy is best understood as a regenerative intervention that may slow progression and promote partial nerve repair — not a guaranteed reversal of all damage.
Does stem cell therapy work for diabetic neuropathy?
Clinical research shows promising results. A 2024 meta-analysis of 7 controlled trials found that stem cell therapy significantly improved motor nerve conduction velocity (WMD +2.2 m/s) and sensory nerve conduction velocity (WMD +1.9 m/s) in patients with diabetic peripheral neuropathy. The Toronto Clinical Scoring System, a measure of overall neuropathy severity, also improved significantly. These objective findings suggest that stem cells may help repair nerve damage in diabetic patients, though larger-scale trials are ongoing.
Is stem cell therapy for neuropathy available in Japan?
Yes. Japan is one of the few countries with a government-regulated framework for regenerative medicine. Under the Act on the Safety of Regenerative Medicine, clinics must register treatment plans with the MHLW and meet strict laboratory and safety standards. Cell Grand Clinic in Osaka offers MHLW-certified stem cell therapy using autologous adipose-derived stem cells, and regularly treats international patients traveling from the United States, Europe, the Middle East, and Asia.
What types of stem cells are used for nerve repair?
Research has explored several types, including bone marrow-derived mononuclear cells (BM-MNCs), umbilical cord-derived mesenchymal stem cells (UC-MSCs), and adipose-derived stem cells (ADSCs). Cell Grand Clinic uses adipose-derived stem cells harvested from the patient’s own fat tissue. ADSCs share the same core regenerative mechanisms — neurotrophic factor secretion, immunomodulation, and exosome release — as other mesenchymal stem cell types, while offering the advantage of being autologous (your own cells, reducing rejection risk) and relatively easy to harvest.
How much does stem cell therapy for neuropathy cost in Japan?
Treatment costs vary depending on the specific protocol, the number of cells cultured, and whether additional therapies are combined. Cell Grand Clinic provides a personalized cost estimate after an initial medical review. For details, contact the clinic directly via WhatsApp or email — inquiries are free of charge.
How long until I notice improvement after treatment?
Most patients do not experience immediate results. Because stem cell therapy works through gradual biological processes — reducing inflammation, promoting nerve regeneration, and restoring blood supply — improvements typically emerge over a period of several weeks to three months following treatment. Some patients report continued improvement for up to six months. A second treatment course may be recommended to strengthen and sustain the response.
Take the Next Step
Living with neuropathy means living with uncertainty — will the numbness spread, will the pain worsen, will your balance continue to decline? If conventional treatments have reached their limit, regenerative medicine may offer a different path forward. Many neuropathy patients also struggle with broader chronic pain — for more on how stem cell therapy addresses pain at its source, see our guide to stem cell therapy for chronic pain.
Cell Grand Clinic’s medical team is available to review your case, discuss whether stem cell therapy may be appropriate for your type of neuropathy, and answer any questions about treatment in Japan.
Reach us directly — WhatsApp and email inquiries are free of charge.
References
- Mauermann ML, Staff NP. Peripheral Neuropathy: A Review. JAMA. 2026;335(3):255-266. https://doi.org/10.1001/jama.2025.19400
- Alizadeh SD, et al. Human studies of the efficacy and safety of stem cells in the treatment of diabetic peripheral neuropathy: a systematic review and meta-analysis. Stem Cell Res Ther. 2024;15(1):442. https://doi.org/10.1186/s13287-024-04033-3
- Wu Z, Huang S, Li S, et al. Bone marrow mesenchymal stem cell and mononuclear cell combination therapy in patients with type 2 diabetes mellitus: a randomized controlled study with 8-year follow-up. Stem Cell Res Ther. 2024;15(1):339. https://doi.org/10.1186/s13287-024-03907-w
- Zhang WJ, Pi XW, Hu DX, Liu XP, Wu MM. Advances and challenges in cell therapy for neuropathic pain based on mesenchymal stem cells. Front Cell Dev Biol. 2025;13:1536566. https://doi.org/10.3389/fcell.2025.1536566
- Claessens AAE, Vriend L, Ovadja ZN, Harmsen MC, van Dongen JA, Coert JH. Therapeutic Efficacy of Adipose Tissue-Derived Components in Neuropathic Pain: A Systematic Review. Bioengineering. 2024;11(10):992. https://doi.org/10.3390/bioengineering11100992
最終更新日:2026.03.28



 Inquiries by Email
Inquiries by Email

